- HIV and AIDS are major global healthcare concerns.
- Advances in anti-retroviral therapy mean that people with HIV infection live longer, which is leading to new health concerns, such as neurological complications.
- Dr Samikkannu Thangavel at Texas A&M University, USA, aims to understand the mechanisms behind cognitive changes in HIV infection.
- His research shows that alterations in gene expression and protein modifications are likely behind the changes seen in the immune cells of the brain.
Every day, 4,000 people become infected with human immunodeficiency virus (HIV), and it remains a leading cause of death in many countries. In 2021, someone died from acquired immune deficiency syndrome (AIDS) every minute, equating to around 650,000 AIDS-related deaths.
Around 75% of people who carry the virus responsible for AIDS, HIV, have access to anti-retroviral treatment. This prevents the condition from progressing and has improved overall life expectancy. However, researchers are still learning about how living with HIV infection can impact health in the long term. A paradigm shift is happening in the research arena towards enhancing the quality of life for people living with HIV infection.

Neurological complications of AIDS
HIV attacks the body’s immune system, including white blood cells. The virus hijacks the cellular machinery and causes epigenetic changes which alter gene expression. HIV infection can also affect cells of the central nervous system, leading to inflammation that can impair brain function. The collection of cognitive changes associated with HIV infection is known as NeuroAIDS.
A paradigm shift is happening in the research arena towards enhancing the quality of life for people living with HIV infection.
One type of a NeuroAIDS disorder is HIV-associated neurocognitive disorder (HAND), which includes symptoms such as behaviour changes, depression, and memory loss. Specialist methods have been used to characterise some of the changes in the brain resulting from HAND and showed that alterations in genes and proteins are associated with metabolic pathways, neurodegenerative disorders, and dementia.
The exact mechanism of HAND is unclear. However, we do know that HIV does not directly infect neurons; instead, it indirectly leads to disrupted glial function (glial cells support and protect neurons), neuronal dysfunction, or death. Despite effective anti-retroviral therapy, around 50% of people with HIV may have HAND.
Epigenetic mechanisms
Dr Samikkannu Thangavel at Texas A&M University, USA, aims to understand more about HAND progression. He explains that HIV may alter the normal functions of cells by changing gene expression – in other words, whether a gene within the cell is turned on or off. When a gene is switched on, this means that the instructions in our DNA can be converted into an end product, for example a particular protein. In particular, Thangavel is interested in an aspect of gene modification called epigenetics.
Epigenetics is a reversible change in the way that genes are read, caused by your behaviours and environment. For example, some epigenetic changes increase the risk of developing cancer, as they alter the way that a certain gene functions. While gene expression may change, the underlying DNA sequence remains the same.
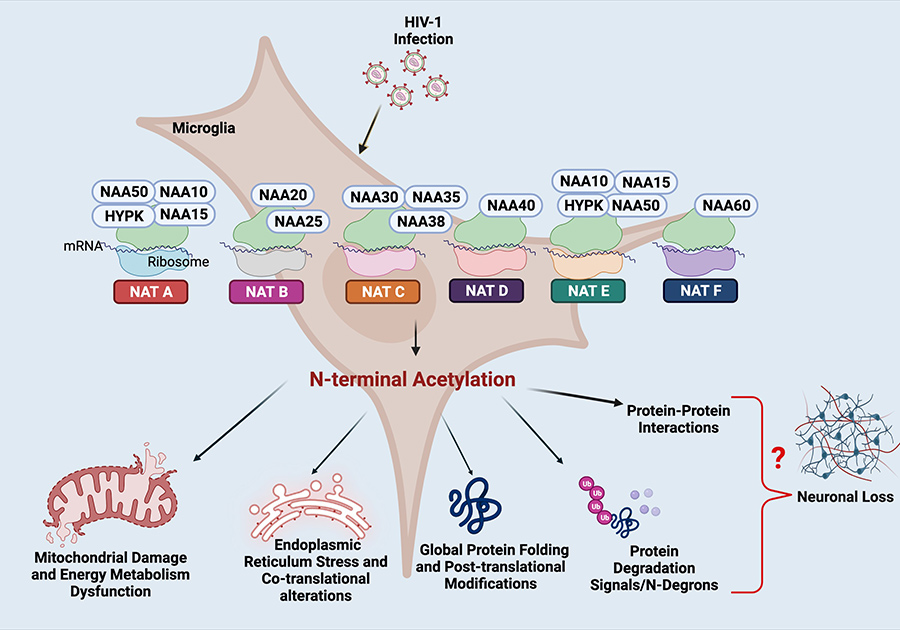
Epigenetic mechanisms can include the addition of a chemical group to one end of a protein, a process called N-terminal acetylation. Protein synthesis within the cell happens in ribosomes, cellular machines that link smaller building bricks together to form larger proteins. Sometimes proteins are altered, or modified, during this stage of production.
HIV can modify immune cells
Thangavel is particularly interested in N-terminal acetylation within an important cell type in the brain, the microglia. Microglia cells are often known as the immune cells of the central nervous system, and play important roles in brain infections, such as HAND. During infection with HIV, the microglia are unable to carry out their normal protective roles and this can promote inflammatory responses within the brain.
Thangavel proposes that N-terminal acetylation changes could explain how the virus and body interact.
Alterations to proteins within the microglia cells through N-terminal acetylation can have a number of consequences, such as impairment of the mitochondrial energy-production and metabolic functions in the microglia, which cause misfolded or degraded proteins, or alterations to signalling pathways within the cell. There may also be issues with oxidative stress and redox imbalance, meaning an imbalance between oxidants and antioxidants. This is likely to affect energy resources and metabolic fuel within the microglia. Many of these changes have already been linked to the development of neurodegenerative disorders.
Research has shown that HIV can interrupt the way these energy powerhouse structures work, including increasing the release of harmful molecules that can contribute to the progression of HAND. In addition, this energy is needed for effective gene expression within the cell. Studies done by Thangavel’s research group at Texas A&M University have already shed light on the epigenetic changes in the human brain cause energy dysfunction, which can contribute to HAND.

Thangavel also suggests that N-terminal acetylation may impact the signalling cascade of gene expression which has been shown to be significantly associated with depressive behaviours, problems with forming memories, and dementia. He proposes that N-terminal acetylation changes induced by viral infections could explain how the virus and body interact. He also explains that if more can be understood about how HIV effects the cell’s energy source, it may be possible to develop therapeutic approaches to prevent HAND, as well as new ways to test for the condition.
Improving quality of life
Further research is still needed to learn more about the relationship between HIV infection, modifications to proteins via N-terminal acetylation or other mechanisms, and cognitive impairment or behavioural changes. It is likely that epigenetic modifications regulate both gene expression in the energy-producing compartments of a cell, as well as gene expression more broadly.
Improved diagnosis and management of cognitive disorders in people with HIV infection, which is well managed through medications, means that overall quality of life can be improved for those living with the virus.
How did you first become interested in the impact of HIV infection on the brain?
Our research focus is aligned with the global efforts to eliminate the suffering of HIV. Understanding the mechanisms of disease progression is key to developing new therapeutic strategies. In the early phase of my research, there was a lack of knowledge of HIV and its virulent subtypes. Our experiments highlighted the higher virulence of the subtypes found in the US compared to European, Asian, and African subtypes, which trigger neuronal cell death through the excess production of a neurotoxin called quinolinic acid. Neurological impairments caused by the subtypes from the US were also somewhat elevated, which paralleled with the high occurrence of HAND in the US.
Disruption of energy homeostasis is one of the features observed in neurodegenerative conditions, including HAND. I was intrigued to investigate the effect of HIV infection on mitochondria – the cell’s powerhouse – and energy metabolic profiling, especially in the epigenome landscape. A series of experiments conducted by our team uncovered the involvement of a variety of epigenetic modifiers, such as piRNA, miRNA, and LINC-RNAs, which were dysregulated in the astrocyte energy reservoirs. We have identified the epigenetic signature in HAND, which has opened new avenues in devising treatment strategies in the arena. Our findings thus would sustain the escalating demand to alleviate the sufferings of patients with HAND.
What do you think will be the next big step in treating HIV/AIDS?
The vaccine has been developed against HIV and is in the FDA approval pipeline. Shortly, we are going to witness an HIV-free world.