There is an explosion in the dietary supplement marketplace fuelled by changes in modern lifestyles. These include a focus on healthy aging and maintaining good health for as long as possible, preventative interventions and therapies, greater attention to personal accountability for health, and advertising strategies. As Brian Huber, Vice President of Therapeutic Areas, Drug Development and Consulting at ICON, explains, the latter is particularly problematic. For the average consumer, the lines are being blurred between drugs and supplements. This is frequently due to misleading marketing ploys and vagaries in legal and regulatory boundaries.
Dietary supplements are very broadly defined by US and EU law as “products taken by mouth that contain dietary ingredients including vitamins, minerals, amino acids, herbs and botanicals, as well as other substances, that can be used to supplement the diet”. These products are available in stores throughout the world, as well as on the internet and are labelled as “dietary supplements”.
Synthetic versions of natural products
Some dietary supplements are used to manage clinical medical conditions, such as vitamin or mineral deficiencies. These deficiencies may result from poor diet quality, restrictive diets or to counter nutritional changes seen with normal aging. For example, calcium and vitamin D supplements may help slow normal bone loss and vitamin B12, essential for red blood cells and nerve cells, may be less effectively absorbed in the aged population. Hence, dietary supplements, when taken in appropriate dosages, can play an important role in health management.
However, many people are unaware that supplements may carry some level of risk when consumed in high amounts, taken for long periods of time, used in combination with certain prescription drugs, or when used without supervision from a qualified healthcare professional. In these situations, dietary supplements may be harmful. The body has very specialist systems for processing and excreting compounds, including dietary supplements. If taken in large doses the body struggles to effectively clear the supplements and this can lead to complications. Certain over-the-counter (OTC) dietary supplements may also carry additional risk. For example, it is not recommended to take some herbal supplements alongside anti-clotting medications, as they can interfere with the action of the medication.

Drugs versus dietary supplements
The main distinction between drugs and dietary supplements is associated with the regulation of health claims. US and EU regulatory authorities have robust review of all safety and efficacy claims related to the treatment, diagnoses, prevention, or cure of diseases. No dietary supplement can make any of those claims. In fact, dietary supplements must overtly maintain that they are not intended to treat, diagnose, prevent, or cure diseases. However, the dividing line between drug claims versus dietary supplement claims has become a very fine line of distinction and is often confusing. In addition, the term “dietary supplement” and “nutraceutical” are often used interchangeably but there is no mention of nutraceuticals in any legislative documents.
“Some dietary supplements are used to manage clinical medical conditions, such as vitamin or mineral deficiencies.”
The World Health Organization (WHO) publishes the International Classification of Diseases (ICD). They are on the 10th revision of this, so it is abbreviated ICD10. ICD10 codes are currently the cornerstone of classifying diseases, injuries and inpatient procedures. Healthcare personnel, such as physicians and nurses, assign ICD-10 codes to verbatim, or abstracted diagnosis or procedure information. ICD-10 codes are also used for billing and claims reimbursement.

Dietary supplements are not intended to treat, diagnose, prevent, or cure any disease. Therefore, dietary supplement manufactures cannot make specific claims as defined by ICD10 codes. As example, Restless Leg Syndrome (RLS) is a very common movement disorder that can affect sleep and quality of life issues. RLS is a recognised and billable “disease / syndrome” and there are several drugs that are approved for use by the FDA for the treatment of moderate to severe RLS. As with all drugs, they went through rigorous regulatory review with subsequent approval, including the FDA approved drug label with specific claims for treatment of RLS.
There are also other OTC dietary supplements available that are marketed to treat RLS, but they cannot use RLS in their specific claims. As they cannot directly claim to treat recognised diseases or syndromes, they tend to use language that alludes to this, without actually making any specific claims; instead, manufacturers use terms such as “calm jumpy legs and promote restful sleep”.

Regulation
The US Food and Drug Administration’s (FDA) responsibilities are similar to those of several other government agencies across the globe, such as the Medicines and Healthcare Products Regulatory Agency (MHRA) in the United Kingdom, the European Medicines Agency (EMA) and the Pharmaceuticals and Medical Devices Agency in Japan.
New drugs must be proven safe and effective and meet the criteria set by regulating bodies before they can be marketed to the public. Manufacturers must also prove they are able to make the drug according to federal quality standards. If a product is approved, it means the agency has determined that the benefits of the product outweigh the known risks for the intended use.
However, regulations do not require dietary supplements to be proven safe to FDA’s or EMA’s satisfaction before they are marketed. FDA assumes drugs are unsafe until proven safe but considers dietary supplements safe until proven unsafe.

rumruay/Shutterstock.com
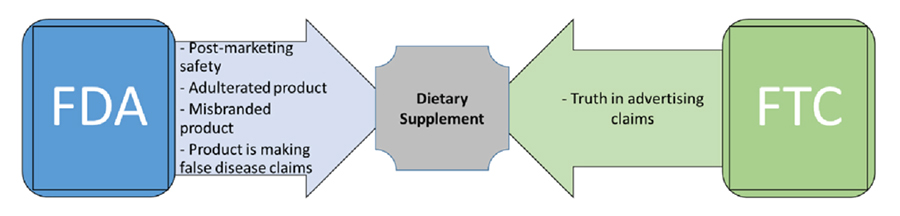
As supplement manufacturers are not required to test their products for safety and/or effectiveness prior to marketing, the FDA’s main role with dietary supplement products begins after the product enters the marketplace. The FDA and EMA will exercise their regulatory authority if the dietary supplement appears to be unsafe, the product is adulterated, the product is misbranded, of if the product is making false claims.
Once a dietary supplement is on the market, FDA has certain safety monitoring responsibilities, such as monitoring adverse events reported by the manufacturer, consumers and healthcare professionals. The FDA has required some supplements to be recalled due to post-marketing safety concerns, contamination and false advertising.

False advertising claims fall under the jurisdiction of the Federal Trade Commissions (FTC). The FTC is the federal agency that regulates many types of advertising and protects consumers by stopping unfair, deceptive or fraudulent practices in the marketplace.
Case study
Dietary supplements often claim to be supported by research. In reality, diligence has shown that statements such as “Clinical proven in clinical trials” may actually have no clinical studies supporting the claims or are linked to very low-quality studies. There are several case studies that can be used as examples of this, including Prevagen, a product marketed to help people with mild memory problems that are frequently associated with aging.
Quincy Bioscience claims that the active ingredient in Prevagen, apoaequorin, is a protein that uniquely supports and improves brain function. Quincy Bioscience states these claims are supported by a double-blinded, placebo-controlled clinical study. Consumers, believing these claims, have spent more than $175 million on Prevagen over the past 10 years. However, there are scientific, clinical and legal issues surrounding this brain supplement.
Apoaequorin, the active protein ingredient in Prevagen, has been used as a research molecule for more than 40 years to study the regulation of calcium flux. It was therefore thought that apoaequorin may help regulate calcium in the brain and thus reduce memory loss and cognitive decline.
Firstly, from a scientific perspective, apoaequorin, a large protein, is not likely to be absorbed orally and will not likely cross the blood-brain barrier.
Secondly, the clinical trial run by Quincy Bioscience “found no overall benefit compared to a placebo for its primary clinical study endpoints involving memory and cognition”. After conducting the analysis that is considered best practice for clinical trials, there was no benefit of Prevagen over placebo. However, Quincy Bioscience went back and did approximately 30 post-hoc (retrospective analysis) tests on the data and a small number of these retrospective post-hoc tests were statistically significant. The post-hoc analysis performed by Quincy would never be accepted in a peer review process as post-hoc tests are usually only used after an initial significant difference between the treatment and control group has been demonstrated.

In 2017, the FTC filed a complaint against the makers of Prevagen for false advertising claims. The FTC charged the maker with falsely advertising that the product improves memory, provides cognitive benefits, and is “clinically shown” to work. According to the FTC, “the marketers of Prevagen preyed on the fears of older consumers experiencing age-related memory loss”. In one of their court appearances, lawyers from Quincy Bioscience admitted they did not “dispute that if you look across the entire 211 people who completed the study there was no statistically significant difference”.
In 2020, a separate class action lawsuit against the manufacturers of Prevagen was settled. The company agreed to partially reimburse people who had purchased the product and to change the claims made on the product label.
“The main distinction between drugs and dietary supplements is associated with regulation of proven health claims.”
While there is a place for some dietary supplements in managing health conditions, they are not indicated for most people and can even lead to further health complications. Dietary supplements should only be taken with input from a healthcare professional who is able to distinguish between the legitimate medical benefit supported by dietary supplements and those making false, misleading and unsubstantiated health claims. Clearly, greater education is needed to help people make informed choices about dietary supplements.

Quality Stock Arts/Shutterstock.com
- (2014). Scrutinising the Term ‘nutraceutical’- a global regulatory perspective [online]. Nutraceutical Business Review. Available at: www.nutraceuticalbusinessreview.com/news/article_page/Scrutinising_the_term_nutraceutical__a_global_regulatory_perspective/100047 [Accessed 06 04 2021]
- Lake, L. (2017). Prevagen’s fishy brainpower claims [online]. US Federal Trade Commission – Consumer Information. Available at: https://www.consumer.ftc.gov/blog/2017/01/prevagens-fishy-brainpower-claims [Accessed 06 04 2021]
- Schardt, D. (2017). Prevagen: How Can This Memory Supplement Flunk Its One Trial and Still Be Advertised as Effective? [online] Center for Science in the Public Interest. Available at: https://cspinet.org/news/prevagen-how-can-memory-supplement-flunk-its-one-trial-and-still-be-advertised-effective [Accessed 06 04 2021]
- (2017). FTC, New York State Charge the Marketers of Prevagen With Making Deceptive Memory, Cognitive Improvement Claims [online]. Federal Trade Commission – Protecting America’s Consumers. Available at: https://www.ftc.gov/news-events/press-releases/2017/01/ftc-new-york-state-charge-marketers-prevagen-making-deceptive [Accessed 06 04 2021]
- (2017). United States District Court Southern District of New York. Complaint for permanent injunction and other equitable relief [online]. Available at: https://www.ftc.gov/system/files/documents/cases/quincy_bioscience_complaint-filed_version.pdf [Accessed 06 04 2021]
- Gastelu, D. (2019). Dietary Supplement Regulations: Comparing US to Canadian model [online]. Natural Products Insider. Available at: https://www.naturalproductsinsider.com/regulatory/dietary-supplement-regulations-comparing-us-canadian-model [Accessed 06 04 2021]
- Marvar, A. (2020). The Prevagen Settlement & the Wild West of Brain Health Supplements [online]. Being Patient. Available at: https://www.beingpatient.com/prevagen-settlement/ [Accessed 06 04 2021]
10.26904/RF-135-1227225972
Contact
Brian Huber

E: Brian.Huber@iconplc.com
T: +1 919 724 5555
W: http://www.ICONplc.com









