Professor Arun Bhunia, Dr Rishi Drolia and the research team at Purdue University, Indiana, USA, explore the mechanisms used by foodborne pathogens when they infect the gastrointestinal tract. They have focused in particular on Listeria monocytogenes, a bacterium that can cause serious illness in high-risk populations. The researchers were able to show a novel pathway used by the bacteria to move from the intestine to the bloodstream. Once they understood more about this mechanism, they were able to use an engineered probiotic to reduce infection rates in mice. This has important implications for the management and treatment of foodborne bacterial diseases.
Scientists have identified over 250 foodborne diseases, most of which are infections caused by a variety of bacteria, viruses, and parasites. One example is listeriosis, an infection that is caused by a bacterium often found in processed meats and dairy products called Listeria monocytogenes. Listeriosis is most likely to affect vulnerable populations, including pregnant women, young children and people with weakened immune systems. The World Health Organization estimates that globally around 23,000 people develop listeriosis each year. Of these, around 5,000 cases are fatal.
Listeria is transmitted from contaminated food. After the food has been ingested, the bacterium crosses the intestinal wall before spreading to other organs of the body including the liver, spleen, brain and placenta (in pregnant women) where it is able to cause further disease.
However, relatively little is known about the mechanisms used by Listeria to cross the intestinal barrier. Understanding more about the way Listeria spreads around the body would allow us to consider strategies for preventing infection in high-risk populations.
At Purdue University in Indiana, USA, Professor Arun Bhunia, Dr Rishi Drolia and the team are researching the mechanisms of pathogenesis (development of disease) used by Listeria to enter the bloodstream.
“The World Health Organization estimates that globally around 23,000 people develop listeriosis each year.”
Epithelial barrier crossing
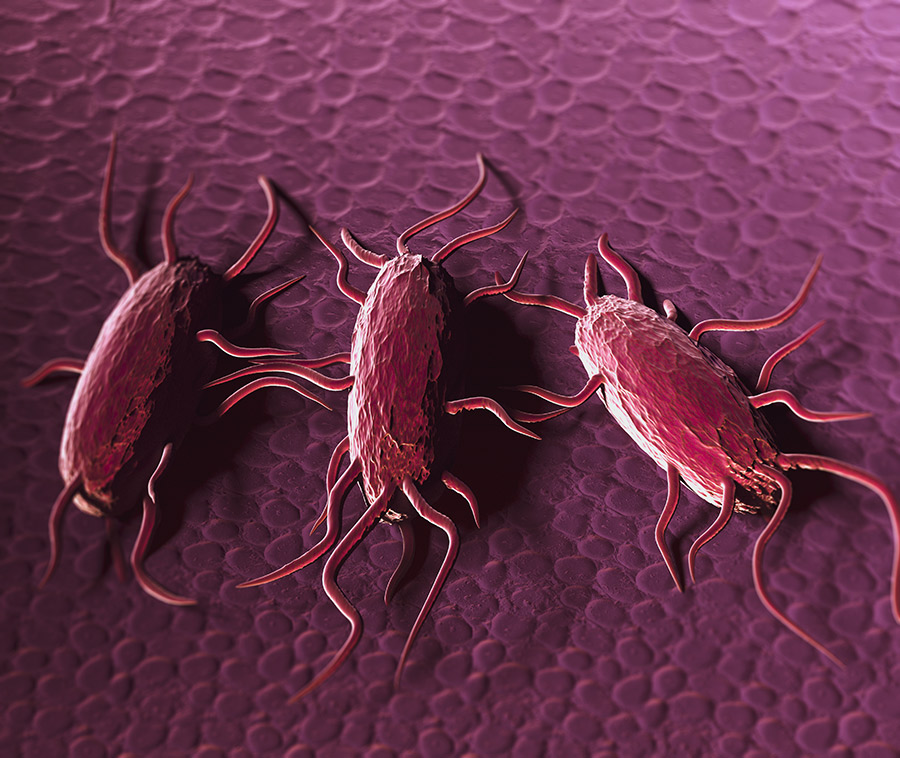
Previously, it was believed that a protein found in Listeria, Internalin A, was necessary for crossing the intestinal barrier. However, research findings from Prof Bhunia and his team shed new light on this. They used mice which had a dysfunctional receptor for Internalin A, meaning that Listeria should not have been able to move from the intestine into the bloodstream and the mice should not have developed listeriosis. After the mice were fed Listeria, the scientists noticed that the bacteria had still been able to cross the intestinal epithelial barrier (see figure 1), suggesting that there was another method the bacteria were using to get into the bloodstream.

Listeria adhesion protein
The research group at Purdue University have discovered a novel pathway that signals to Listeria to begin the process of travelling from the intestine to other body tissues. This happens around 12-48 hours after infection. Prof Bhunia and his team identified a virulence factor – a molecule produced by the bacteria that affects its ability to cause infection – that promotes pathogenesis of Listeria, called Listeria adhesion protein (LAP).
This protein promotes the attachment of the Listeria bacterium to cells in the intestinal wall and helps it pass through the intestinal barrier, which is made up of epithelial cells, a type of cell that forms a covering for all body surfaces. The intestinal barrier allows the body to absorb essential nutrients from the intestinal contents, whilst preventing harmful microorganisms from passing through from the intestine wall to enter the body.
The researchers also found that LAP interacts with another protein: heat shock protein 60 (Hsp60). Using animal and laboratory cell models, Prof Bhunia and Dr Drolia showed that interactions between the two proteins initiate a cascade of cell signalling events that lead to the activation of another well-known signalling pathway, called the NF-kB signalling pathway. The NF-kB pathway involves a further cascade of events that accumulate to control cell survival, DNA processing, and production of other cell signalling molecules.
One outcome of this pathway is changes in the intestinal barrier, mediated by an enzyme known as myosin light chain kinase (MLCK). Usually, junctions between neighbouring cells ensure the integrity of the intestinal barrier. However, changes induced by LAP mean that these junctions are not as watertight as normal, and the cells move apart. This means that bacteria such as Listeria can move from the intestine into the bloodstream and translocate to other organs in the body.
Bioengineered probiotics
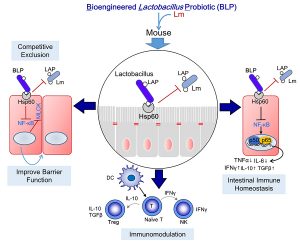
Once they understood more about how the LAP-Hsp60 interaction contributed to the pathogenesis of Listeria, Prof Bhunia, Dr Drolia and the team were able to develop a probiotic that could prevent this interaction. They used a bacterium known to be beneficial for gut health, Lactobacillus, and engineered the Lactobacillus to express LAP proteins on its surface. The resulting probiotic was named bioengineered lactobacillus probiotic (BLP). This meant that the probiotic bound to host Hsp60 proteins, reducing the number of binding sites for Listeria and thus preventing the pathogenic bacterium from entering the bloodstream.
Following 10 days of probiotic treatment, the BLP showed around a tenfold higher level of colonisation in the intestine compared to the control probiotic which had not undergone any engineering steps. In addition, the BLP did not seem to be able to cross the intestinal barrier, showing that it is unlikely to cause any health complications.
Saiful52/Shutterstock.com
Using a mouse model of Listeria infection, the scientists were able to show that the Lactobacillus probiotic successfully colonised the intestine for a week or longer, reduced adherence of Listeria to the intestinal wall, and subsequently reduced the spread of the bacterium to other areas of the body. Indeed, the amount of Listeria bacteria that adhered to the intestinal wall was reduced by about 100-fold in mice that received BLP, compared to mice in the control group. Even bigger reductions were seen in the number of Listeria shed in the faeces of the mice, suggesting a lower bacterial burden in the mice who received the BLP. Overall, the study found that BLP protected mice from a lethal listeriosis infection in 92% of cases.
“There may be opportunities to use the same technologies and methodologies to create novel probiotics.”
The research group also carried out a study to further investigate the mechanisms by which the probiotic was able to provide protection from listeriosis. The scientists hypothesised that BLP led to the formation of co-aggregates – or attachments to Listeria cells – and that this meant Listeria was unable to bind to Hsp60 and exert its effect on the intestinal barrier.
The results of the trial demonstrated that the effects of the probiotic were due to improved intestinal epithelial barrier function, promotion of effective immune responses in the intestine, and direct competition for binding sites which led to exclusion of Listeria interactions with intestinal barrier molecules (see figure 2). Their earlier hypothesis was correct: LAP did promote the formation of co-aggregates between Listeria, the BLP and the LAP protein itself.
There are many health benefits which have already been reported for probiotics: they are often taken after a course of antibiotics, for example, or used to improve symptoms of irritable bowel syndrome. The findings of the study undertaken by Prof Bhunia and his team add another benefit of probiotics to the list and represent a new paradigm for pathogen exclusion. The study is particularly important as a major limitation for therapeutic probiotic use is often the probiotic’s poor ability to colonise the intestine, an issue that was successfully overcome in this trial. There may be opportunities to use the same technologies and methodologies to create novel probiotics to treat other intestinal conditions caused by bacteria. In addition, Prof Bhunia’s team has begun developing therapeutics that block LAP function, which would offer an alternative method for treating listeriosis.

Limiting the spread of infection
The majority of listeriosis cases are due to the consumption of contaminated food. Prof Bhunia and Dr Drolia explain, therefore, that restricting Listeria when it is present in the intestine is the best time to limit the spread of the bacteria to other tissues of the body, where infection can be fatal.
Their data suggest that there is a need to rethink the mechanisms that Listeria uses to infect humans. Rational engineering of probiotics significantly improves their ability to modulate immune response to infection, as well as directly interfere with adhesion to the intestinal epithelial barriers. Taken together, this represents a new paradigm for the treatment of listeriosis and offers an approach with favourable regulatory compliance.
Is there a risk of bacteria such as Listeria developing resistance or ways to evade engineered probiotics, as there is with antibiotics?
The engineered probiotic-mediated anti-Listeria effect is multifaceted; thus, there is very little chance for developing resistance against listeriosis. BLP occupies the host receptor, Hsp60, thereby preventing Listeria from interacting with the host intestinal cells. Intimate BLP attachment and colonisation on epithelial cells also promote enhanced epithelial barrier integrity, reduce inflammatory responses and modulate immune response. This provides robust protection against listeriosis. Unlike antibiotics, probiotics do not seem to alter target gene expression in pathogens
which normally happens during antibiotic resistance.
References
- Drolia, R., Amalaradjou, M., Ryan, V., Tenguria, S., Liu, D., Bai, X., Xu, L., Singh, A. K., Cox, A. D., Bernal-Crespo, V., Schaber, J. A., Applegate, B. M., Vemulapalli, R., & Bhunia, A. K. (2020). Receptor-targeted Engineered Probiotics Mitigate Lethal Listeria Infection. Nature Communications, 11(1), 6344. Available at: https://doi.org/10.1038/s41467-020-20200-5
- Drolia, R., & Bhunia, A. K. (2019). Crossing the Intestinal Barrier via Listeria Adhesion Protein and Internalin A Trends in Microbiology, 27(5), 408–425. Available at: https://doi.org/10.1016/j.tim.2018.12.007
- Drolia, R., Tenguria, S., Durkes, A. C., Turner, J. R., & Bhunia, A. K. (2018). Listeria Adhesion Protein Induces Intestinal Epithelial Barrier Dysfunction for Bacterial Translocation. Cell Host & Microbe, 23(4), 470–484.e7. Available at: https://doi.org/10.1016/j.chom.2018.03.004
10.26904/RF-136-1450702539
Research Objectives
Professor Arun Bhunia and Dr Rishi Drolia’s research shows how foodborne pathogens move from the intestine into the bloodstream.
Funding
The U.S. Department of Agriculture (Agreement No. 59-8072-6-001), the Showalter Trust, the USDA National Institute of Food and Agriculture, the Indiana Clinical and Translation Sciences Institute (NIH- sponsored), BioMatrix, Inc., United Animal Health, and Purdue University supported this research.
Collaborators
- Prof Bruce Applegate, Purdue University
- Dr Abigail Cox, Purdue University
- Dr Gregory Knipp, Purdue University
- Prof Sophie A. Lelièvre, Institut Cancerologie Ouest (France)
- Dr Nicholas Noinaj, Purdue University
- Dr Rahim Rahimi, Purdue University
- Prof Jerrold R Turner, Harvard Medical School
- Prof Ramesh Vemulapalli, Texas A&M University
Bio

Arun Bhunia, BVSc, PhD is Professor of food microbiology in the Department of Food Science and is also affiliated with the Department of Comparative Pathobiology, Purdue Institute of Inflammation, Immunology and Infectious Disease, and Purdue University Life Science program. His expertise is in the area of microbial pathogenesis (host-pathogen interaction), probiotic bioengineering, and foodborne pathogen detection.

Rishi Drolia, MS, PhD is a Postdoctoral Research Associate with research interest in bacterial pathogenesis, probiotic bioengineering, host-pathogen interactions, and cell biology.
Contact
Department of Food Science
Department of Comparative Pathobiology (Courtesy)
Purdue Institute of Inflammation, Immunology and Infectious Disease
Purdue University
West Lafayette, IN, 47907, USA

E: bhunia@purdue.edu
E: rdrolia@purdue.edu
T: +1 765-494-5443
W: https://ag.purdue.edu/foodsci/Pages/profile.aspx?strAlias=bhunia