The mammalian inner ear is a labyrinth of incredibly intricate sensory structures. The cochlea, a spiral-shaped cavity, is a vital part of the auditory system and converts vibrations (produced by sound waves) into electrical impulses which communicate to the brain via the auditory nerve.
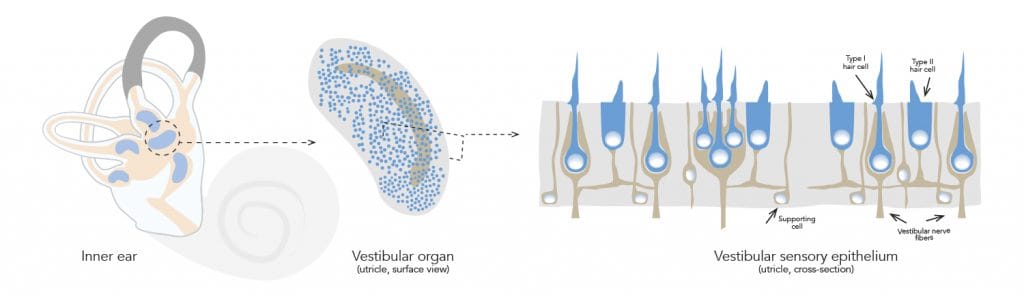 The vestibular system, also part of the inner ear, is essential in maintaining spatial orientation and balance. It is composed of three semi-circular canals, each orientated in a different plane. Within each canal, fluid moves in response to head motion, triggering tiny vestibular hair cell receptors which send signals to the brain and eyes via the hair cells’ innervating neurones. This mechanism gives mammals a sense of balance and coordination. Vestibular hair cells can be further divided into two subcategories: type I and type II. Although knowledge is limited, research suggests that type I hair cells are better suited to detect high-frequency movements than type II hair cells.
The vestibular system, also part of the inner ear, is essential in maintaining spatial orientation and balance. It is composed of three semi-circular canals, each orientated in a different plane. Within each canal, fluid moves in response to head motion, triggering tiny vestibular hair cell receptors which send signals to the brain and eyes via the hair cells’ innervating neurones. This mechanism gives mammals a sense of balance and coordination. Vestibular hair cells can be further divided into two subcategories: type I and type II. Although knowledge is limited, research suggests that type I hair cells are better suited to detect high-frequency movements than type II hair cells.Vestibular System Degeneration
The hair cells and innervating neurones associated with the vestibular organ are very vulnerable to degeneration, which increases with age. In fact, approximately 35% of the United States population aged 40 or over are affected by some form of vestibular system disorder and the consequences can be devastating. Patients may experience debilitating bouts of intense vertigo and imbalance. In addition, sufferers often find it difficult to concentrate and physically struggle to perform routine activities, causing great emotional distress.
Understanding the molecular background underpinning hair cell differentiation and regeneration is vital if we are to develop therapies to treat vestibular disorder![]()
Current options for treating vestibular disorders are limited and include vestibular rehabilitation therapy, medication and surgery. Even in a best-case scenario, symptoms are only improved, not cured. The only way in which vestibular function could be fully restored would be via hair cell regeneration and subsequent nerve innervation.
Dr Stone and her team are currently exploring this process in depth and hope to develop more effective therapies to treat vestibular disorders via cellular regeneration.
Hair Cell Regeneration
Non-mammalian animals respond to vestibular damage by regenerating both type I and type II hair cells. In birds, amphibians, and fish, new hair cells arise via mitosis (cell division) of supporting cells and their subsequent differentiation into hair cells. Replacement hair cells in non-mammals are also produced via direct transdifferentiation – a non-mitotic process whereby supporting cells are phenotypically converted into hair cells. In birds, studies have shown that regenerated cells become innervated, restoring function.
However, in a recent study, Dr Stone and colleagues showed that only type II vestibular hair cells can regenerate in mammals. The team destroyed vestibular hair cells in adult mice by inserting the destructive human diphtheria toxin receptor (DTR) gene into the locus for the Pou4f3 gene. Sixty days after treatment, they discovered that hair cell numbers had actually increased significantly, despite little increase in mitotic activity, suggesting that regeneration had occurred via the direct transdifferentiation of supporting cells (rather than mitosis).
Intriguingly, however, there is no evidence to suggest type I hair cells are replaced and Dr Stone and her team are now investigating the properties of type I hair cells to understand whether they can also regenerate.
Type II Hair Cells’ Unique Properties
In order to distinguish some of the differences between the two hair cell types, Dr Stone and her collaborator Rémy Pujol used confocal and transmission electron microscopy (TEM) to study hair cell structure in adult mice. Unlike type I hair cells, type II hair cells have basolateral processes (processing units) that are in physical contact with each other, forming a delicate network. However, more research is needed to explain this unusual phenomenon – perhaps the connection is simply for mechanical support, or maybe it enhances communication between hair cells.
A more radical idea is that a direct link between the hair cell population could regulate their homeostasis. Dr Stone, in collaboration with Brandon Cox’s lab at Southern Illinois University School of Medicine, has attained evidence that vestibular type II hair cells undergo ‘turnover’ under normal conditions: individual hair cells are culled from the sensory organs and then replaced via transdifferentiation of supporting cells. In contrast to normal conditions, hair cell destruction causes supporting cells to produce six times the amount of replacement type II hair cells. This plasticity (adaptability) of vestibular organs may help to ensure balance function is retained in adult mammals, perhaps even in humans.
Approximately 35% of the United States population aged 40 or over are affected by some form of vestibular system disorder![]()
Molecular Basis of Hair Cell Regeneration
Understanding the molecular background underpinning hair cell differentiation and regeneration is vital if we are to develop therapies to treat vestibular disorders.
The ‘Notch signalling pathway’ is particularly important in the process of hair cell development in embryos. Dr Stone and her team have shown that this pathway inhibits the production of a key basic helix-loop-helix transcription factor that activates hair cell differentiation, called atonal homolog 1 (Atoh1).
During embryo development, signalling proteins bind to the ‘Notch’ receptor, located on undifferentiated cells, activating enzymes which cleave the receptor. Subsequently, the cleaved protein activates genes that encode other proteins that inhibit Atoh1.
To determine whether Atoh1 is reactivated after vestibular hair cell loss, Dr Stone and her team performed a study on adult mice utricles (an organ found in the vestibular system), in which hair cells had been destroyed using neomycin. Interestingly, the team detected Atoh1 expression in the supporting cells 4 days after neomycin treatment. These supporting cells then underwent direct transdifferentiation to form very primitive hair cells.
Additionally, Dr Stone showed Notch pathway inhibition resulted in an increase in Atoh1 levels, and supporting cells progressed to later stages of hair cell differentiation. Again, however, these new hair cells were not fully functional – they lacked hair bundle maturation and innervation. Dr Stone is working with a consortium of international scientists, the Hearing Restoration Project, funded by the Hearing Health Foundation, to determine additional signals that regulate hair cell regeneration in adult mice.
Future Research
Dr Stone’s research is extremely promising, indicating that mammalian adult hair cells have the potential to regenerate via the phenotypic conversion of supporting cells. However, many questions remain unanswered. For example, what are the molecular processes involved in regulating hair cell maturation? Are there ways in which we can initiate type I hair cell regeneration? And, what molecular mechanisms underpin vestibular cell diversity?
Overcoming these obstacles will take us one step closer to developing hair cell replacement therapies used to treat vestibular disorders and improve the lives of thousands of sufferers.
Since my graduate training, I have studied auditory and vestibular hair cell regeneration in birds. I wanted to move my lab’s research into a model system that was more like humans, and the best option was mice, since they offer abundant genetic tools. There was already some evidence that replacement of vestibular hair cells might happen in adult guinea pigs, but it was clear it did not occur in the cochlea. We knew little about vestibular hair cell regeneration in mice at that time. This background, coupled with the knowledge that understanding more about hair cell regeneration in mammals could someday help people with vestibular dysfunction, motivated me to work in this area.
Why do vestibular hair cells degenerate with age?
We don’t know the answer to this. However, hair cells are highly metabolically active all of the time; they never rest! So, they are probably more susceptible to wear and tear than other cell types.
Why do non-mammalian hair cells have a higher level of plasticity, compared to mammalian?
We wish we knew! Regeneration of multiple tissue types is an enviable property among lower vertebrates, and the predominance of regenerating tissues in a given animal seems to be inversely proportionate to its morphological complexity.
Is there a chance that we could manipulate the molecular pathways that regulate hair cell regeneration for clinical benefit?
We certainly hope so! Research advances in our field each year bring us closer to the identification of molecules that have significant and lasting effects on hair cell replacement in mammals.
Where do you see your research progressing in 5 years’ time?
I hope that we will identify ways to promote replacement of higher numbers and both types of vestibular hair cells in mice, such that balance function is restored in a meaningful and lasting manner. This will be a key step in moving toward a clinical application for our research.
Dr Stone’s research focuses on the replacement of vestibular hair cells in mature rodents as a possible means to learn more about cell death in humans and the possibility for regeneration.
Funding
National Institutes of Health; the Hearing Health Foundation; the Virginia Merrill Bloedel Hearing Research Center
Collaborators
- Members of the Hearing Restoration Project, funded by the Hearing Health Foundation
- The Virginia Merrill Hearing Research Center, University of Washington
- Edwin Rubel, University of Washington
- James Phillips, University of Washington
- Brandon Cox, Southern Illinois University
- Ruth Anne Eatock, Chicago University
- Rémy Pujol, University of Montpellier, France
Bio
Contact
Jennifer Susan Stone, PhD
Research Professor
Dept of Otolaryngology
VM Bloedel Hearing Research Center
CHDD CD 176
Box 357923
University of Washington
Seattle, WA 98195-7923
E: stoner@uw.edu
T: +1 206 616 4108 or 206 616 4155
W: http://faculty.washington.edu/stoner/Stone_Lab/Home.html
http://otolaryngology.uw.edu/faculty/jennifer-stone









