Technology that uses magnetic force has the potential to help in a variety of medical settings. Dr Michael Harrison and his colleagues have developed and tested titanium enclosed permanent magnets that are surgically implanted using minimally invasive techniques to address unmet clinical needs.
One device called MagNap helps to maintain an open airway at night for patients with obstructive sleep apnoea. A second device Magimplant/Magnatrack, part of the Magnetic Mini-Mover Procedure, helps to repair an anatomic malformation called pectus excavatum (or sunken chest) in children aged between eight and 13. Another device named Magnamosis surgically creates a stronger, leak-free, post-surgery connection (anastomosis) in the gastrointestinal or biliary tract. His team is also working on utilising the Magnamosis device for the surgical treatment of type 2 diabetes. Finally, his device innovation team have worked on the development of magnetically controlled growing rods called Roboimplant to treat scoliosis and assist in the process of limb lengthening.
Magnetic force to change anatomic development
Magnetic fields created by permanent rare earth magnets or by electromagnets are behind a number of technological tools present in the world today. These include computer storage, speakers and headphones, and Magnetic Resonance Imaging (MRI) scans, which allow doctors to examine patients’ bodies internally, in order to spot abnormalities and diagnose health conditions. But Dr Harrison’s devices use the force generated by the magnetic fields to gradually alter the development and biology of anatomic structures.
Dr Harrison is an internationally renowned scientist, primarily known for his work in paediatric and foetal surgery. While researching aspects related to foetal surgery, the professor and his team invented tools and devices that made foetal intervention possible. Dr Harrison continued to invent and develop new tools that could tackle unmet clinical needs even after retiring from clinical practice. His most recent work focused on developing devices that use the magnetic force to surgically treat sleep apnoea (MagNap) and another to replace staples for intestinal anastomotic connections (Magnamosis).
A magnetic implant to correct pectus excavatum: magnetic mini-mover
The first device Dr Harrison developed addresses pectus excavatum deformities, or sunken chest. This is the most common congenital chest wall defect, which is characterised by a depression at the back of the sternum. About one in 400 children are born with this disorder and over 10,000 require the surgery worldwide. Dr Harrison was not satisfied with standard surgical interventions for pectus excavatum including the Nuss and Ravitch procedures which attempt to correct the skeletal defect in one big procedure. He instead took the approach that a little pressure applied over a longer time could remodel the abnormal cartilage.
Magnetic force has the exciting potential to simplify a number of complex and tricky surgical procedures![]()
The procedure devised by Dr Harrison and his team, called magnetic mini-mover procedure (3MP), works by applying a sustained outward force on the depressed sternum in order to correct its anatomical position. One titanium-encased magnet is surgically implanted on the front of the sternum via a 2cm incision, performed as an outpatient under general anaesthesia. A second magnet is worn in a brace on the patient’s chest. By adjusting the strength of the second magnet, the two devices are pulled together, slowly repairing the defect.
A magnetic implant to treat obstructive sleep apnoea: MagNap
The magnetic technology invented to address pectus excavatum inspired Dr Harrison to devise another similar device to treat obstructive sleep apnoea (OSA). Millions of people worldwide suffer from this disorder. Symptoms of OSA include excessive daytime sleepiness, loud snoring, breathing cessation during sleep, difficulty concentrating during the day, mood changes, and abrupt awakenings during the night. OSA can also cause cognitive and behavioural problems, and has been associated with cardiovascular disease and metabolic syndrome. The primary measurement for OSA is the Apnea Hypopnea Index (AHI) which shows the severity of the patient’s problem. Conventional treatments for OSA include positive airway pressure therapy (CPAP) or palate surgical procedures, which can in some instances create disruptions in normal functions such as speech or swallowing. In many cases, the CPAP machine can offer significant relief. However, many patients cannot tolerate the device and the positive pressure mask that must be worn at night. Some patients get no relief from these devices and must resort to a variety of surgical and non-surgical alternatives.
 As an alternative treatment, Dr Harrison and his team demonstrated that pulling the hyoid bone and all its attachments forward opens the collapsing airway in OSA. To achieve this, they implant a titanium enclosed rare earth magnet on the hyoid bone in an outpatient procedure. At night, the patient or partner puts on a small, adjustable external brace that holds a second magnet which interacts with the implanted magnet to open the airway. The external device is worn only while sleeping and by pulling the hyoid directly forward helps to counteract airway collapse, ensuring that patients’ airways remain unobstructed while they sleep.
As an alternative treatment, Dr Harrison and his team demonstrated that pulling the hyoid bone and all its attachments forward opens the collapsing airway in OSA. To achieve this, they implant a titanium enclosed rare earth magnet on the hyoid bone in an outpatient procedure. At night, the patient or partner puts on a small, adjustable external brace that holds a second magnet which interacts with the implanted magnet to open the airway. The external device is worn only while sleeping and by pulling the hyoid directly forward helps to counteract airway collapse, ensuring that patients’ airways remain unobstructed while they sleep.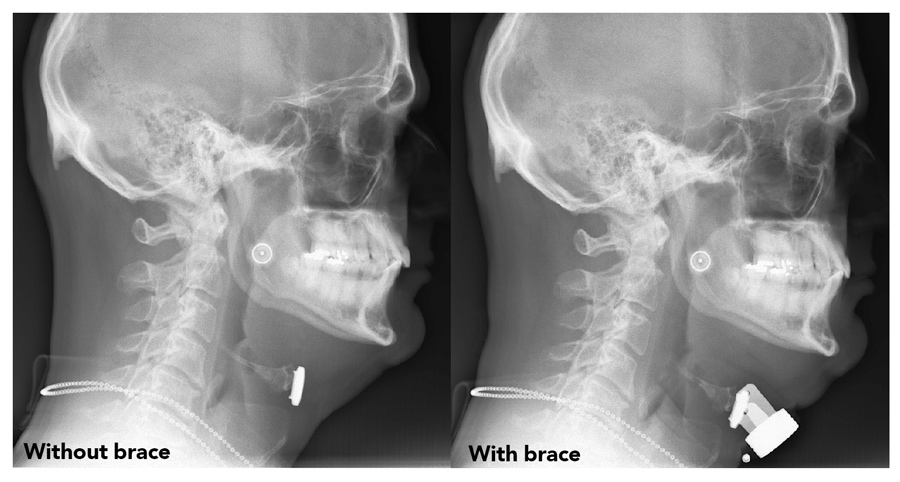
A magnetic device to create intestinal anastomoses : Magnamosis
Magnamosis is a third surgical device invented by Dr Harrison. The device consists of two magnetic rings that helps to create new connections in the gastrointestinal tract in patients with colon cancer, bowel blockages or other procedures that require the joining of two pieces of intestinal tissue. Traditionally, anastomoses are surgically hand-sewn, although more recently the use of staplers has also become common, as they shorten the time of the procedure. Yet both these methods leave foreign objects at the new connection which can cause inflammation and other issues. Dr Harrison’s Magnamosis device consists of two self-aligning magnetic rings covered in a polycarbonate casing. Its geometry facilitates the tissue’s healing and remodelling process. Over time (three to five days) the tissue necroses and a perfect connection remains. Magnamosis was initially tested on pigs and monkeys. Burst testing has shown that the anastomosis created is stronger than either staples or sutures. UCSF is directing a ten-patient FDA-approved clinical trial. Five patients have completed the surgery and a perfect anastomosis was formed in each case. UCSF is actively recruiting patients for this trial. Additional information can be obtained here: https://clinicaltrials.gov/ct2/show/results/NCT02043392.
Dr Harrison continues to invent and develop new tools that could tackle unmet clinical needs even after retiring from clinical practice![]()
Dr Harrison and his team is developing a sensor for the Magnamosis device. This will ensure that the connection between the two magnets is in perfect alignment and exerting sufficient pressure to cause the creation of the anastomosis regardless of the thickness of the patient’s intestinal tissues.
Dr Harrison has received funding from the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) to pursue another potential use of Magnamosis: the treatment of type 2 diabetes. During bariatric surgery, the patient often sees a reversal of type II diabetes before weight loss actually begins. There appears to be a mechanism in the small intestine where certain pulpy acidic fluids (called chyme) are diverted through a specific part of the intestine causing a drop in haemoglobin A1C and reversing the metabolic syndrome. Pre-clinical testing over the next year may show exactly where this change takes place and how much chyme is needed to reverse the diabetes.
The future of magnetic force
Dr Harrison’s work with magnetic devices shows very promising results and has potential applications across a wide range of medical conditions. The magnetic devices for pectus excavatum and OSA could bring considerable advantages for patients affected by these conditions, introducing a less invasive and more tolerable treatment strategy. The Magnamosis procedure could have important implications for reconstructive surgery and can also be applied to the creation of anastomosis in other parts of the body, including the biliary and urinary systems. A new magnetic device called Roboimplant could also be used to correct spinal deformities such as scoliosis, a possibility that Dr Harrison is already investigating. In future, magnetic force has the exciting potential to simplify a number of complex and tricky surgical procedures, performed on both adults and children.
All this work is being developed in partnership with Magnamosis Inc. a device development corporation in San Francisco, California.
The first unsolved problem was how to apply outward force on a sunken chest without having a screw or wire piercing the skin. Magnetic attraction between an implanted magnet and a second magnet in an external brace was the answer.
What do you believe are your most promising findings so far, in terms of applications of the magnetic devices invented by you?
Aside from offering potential solutions for a variety of rather uncommon paediatric surgical problems like oesophageal atresia, there are two potential solutions to very big common problems: Magnap for obstructive sleep apnoea and Duodenal-Ileal bypass (DIPASS) and the Magnamosis device for type 2 diabetes.
The Roboimplant device has been developed, undergone
initial testing, and is being further developed and commercialised
by a start-up company![]()
What are the potential advantages of using magnetic devices compared to standard medical procedures?
In all the conditions we have studied, the use of magnetic force appears to be better, faster, and considerably cheaper.
Have you already started developing a device that can help correct spinal deformities? If yes, what stage of development is it at?
The Roboimplant device has been developed, undergone initial testing, and is being further developed and commercialised by a start-up company.
What are your plans for future research and investigation?
Our team will just keep innovating. It’s fun!
For thirty years, the Harrison lab has developed and tested foetal surgery techniques, first in animal models and then in human clinical trials. The lab now focuses on device innovation, and most recently is exploring the use of magnetic force to correct a variety of surgical problems and deformities. The team has learned that force applied by implanted magnets can over time correct skeletal deformities, create intestinal anastomoses, and even treat obstructive sleep apnoea.
Funding
National Heart, Lung and Blood Institute (NHLBI): National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK); FDA-funded Pediatric Device Consortium; Magnamosis, Inc; Magnap, Inc
Collaborators
Dillon Kwiat; Claire Graves, MD; Jill Imamura-Ching, RN; Richard Fechter; Peter Havel, PhD, DVM; Michael Danty
Bio
 Dr Harrison is Professor of Surgery, Pediatrics, and Obstetrics, Gynecology and Reproductive Sciences, Emeritus at UCSF, where he is Director of the Pediatric Device Consortium. He received his MD with distinction from Harvard Medical School. Acknowledged around the world as the “Father of Fetal Surgery”, Dr Harrison is internationally renowned for his expertise and innovation in paediatric and foetal surgery. A founder of the International Fetal Medicine Surgery Society, and founder and director of the UCSF Fetal Treatment Center.
Dr Harrison is Professor of Surgery, Pediatrics, and Obstetrics, Gynecology and Reproductive Sciences, Emeritus at UCSF, where he is Director of the Pediatric Device Consortium. He received his MD with distinction from Harvard Medical School. Acknowledged around the world as the “Father of Fetal Surgery”, Dr Harrison is internationally renowned for his expertise and innovation in paediatric and foetal surgery. A founder of the International Fetal Medicine Surgery Society, and founder and director of the UCSF Fetal Treatment Center.Contact
Michael R Harrison, MD
Professor, UCSF School of Medicine
3333 California Street
San Francisco CA 94104
USA
E: michael.harrison@ucsfmedctr.org
T: +1 415 476 4914
W: http://profiles.ucsf.edu/michael.harrison









