Periods of dormancy or hibernation are used by many species to avoid harsh environmental conditions. But how does this work in bacteria? Dr Sariqa Wagley, University of Exeter, investigates how Vibrio parahaemolyticus are able to enter a dormant, or viable but not culturable phase, before being resuscitated once conditions become more favourable. Understanding more about this process and ways to detect dormant bacteria has important implications for UK food safety strategies.
In the UK, mean annual sea temperatures have risen by just under 1oC since 1870 and this increase is set to continue at an even faster rate over the coming years, depending on greenhouse gas emission levels. This change in sea temperature has significant impacts on the organisms that rely on it to survive; for example, the population size of large fish looks set to decrease. But it is not just the larger species that will be affected. Dr Sariqa Wagley at the University of Exeter explores how warming sea temperatures in the UK are affecting the increased incidence of bacterial Vibrio species that live in or like to inhabit seafood.
During the winter months, Dr Wagley reports that it is not possible to detect Vibrio bacteria in marine environments. Still, they re-appear in summer and cause infections in humans once more. Increasing sea temperatures may influence how long the bacteria spend in their dormant phase, and how quickly they can be resuscitated and regain the ability to cause disease.
One example of a bacteria often associated with causing seafood-borne gastroenteritis is Vibrio parahaemolyticus.
This hibernation strategy is used by many organisms in the natural world as it presents a way in which they can survive harsh environmental conditions. However, relatively little is known about this dormant state in microbiology; this presents a major barrier to detecting the bacteria as it may represent a reservoir of bacteria that cannot be observed but that can be re-activated later under more favourable conditions.
The research group at the University of Exeter are interested in learning more about the bacterial dormancy phase and how it is used by the bacteria to survive unfavourable conditions during winter.

Bacterial dormancy
Bacterial dormancy, also known as a viable but non-culturable (VBNC) state, has already been described in over 80 other bacterial species. It is a condition when bacterial cells are viable and metabolically active but are resistant to being grown in a laboratory in their usual growth media. Environmental factors known to induce VBNC cells include nutrient restriction, extreme temperatures, and exposure to ultraviolet light. In Dr Wagley’s study, restricting nutrient availability and decreasing the temperature was used to induce a VBNC state in the bacterial cells.
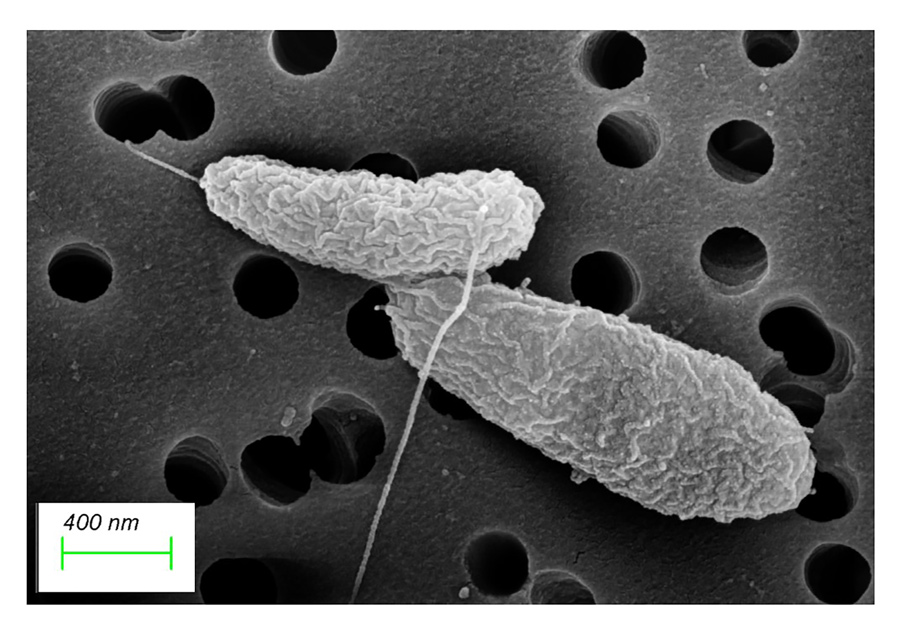
V.parahaemolyticus usually appears as a rod-shaped bacterium which can be detected using routine microbiological techniques in the laboratory. However, in its dormant stage the bacterium changes to a coccoid shape, meaning that it becomes spherical. Furthermore, the bacteria lose their cell contents, i.e. the proteins and other molecules usually used for daily activities such as respiration and metabolism, and they cannot be detected using routine testing.
Using advanced microscopy techniques, Dr Wagley has been able to identify and characterise dormant, VBNC bacterial populations. The research involved investigating the ability of V.parahaemolyticus to form VBNC cells and their potential to be resuscitated from this state. Dr Wagley has then been able to explore the protein content of these cells to try and characterise ways in which the bacteria can survive during this dormant state.
Methods
Dr Wagley used a method called fluorescence activated cell sorting, or FACS, which is a type of flow cytometry that allows scientists to rapidly and accurately collect information relating to a mixture of different cell types. FACS works by tagging fluorescently labelled antibodies onto specific receptors on a cell surface and the amount of light emitted or refracted from the cells is collected by special sensors. The machine is then able to build up a picture of the different populations of cells within a sample, based on what molecules they have on their cell surface.
As well as FACS, other laboratory techniques were used to differentiate bacterial subpopulations based on their metabolic activity, cell shape and ability to cause disease in wax moth larvae. As studies using the native hosts of the bacteria are costly and difficult to practically carry out, the larvae provide a suitable host to study the pathogenicity and dormant state of V.parahaemolyticus.

Two key subpopulations
Dr Wagley found that under unfavourable conditions, most of the bacterial cells died and only around 1% entered the VBNC state. So what was it about this 1% of cells that made them different from other cells?
Firstly, the results of the study showed that usually, rod-shaped cells transform to a more spherical shape in the VBNC state, and that these spherical cells can be resuscitated for up to 14 days after becoming VBNC.
In addition, the findings suggested that there are two key subpopulations of V.parahaemolyticus dormant cells, which the researcher called P1 and P2. Subpopulation P1 appeared as smaller coccoid cells, whereas P2 appeared as larger, more dense coccoid cells that were sometimes present as pairs. Of the 1% of cells that enter the VBNC state, 90% could be classified as belonging to the P1 population whereas only 10% became the larger coccoid form seen in the P2 subpopulation.

Photo Source: Bioimaging centre at University of Exeter
While both of these subpopulations can remain dormant in the VBNC state for long periods of time, P2 was better able to survive stressful conditions over a longer period of time (up to 50 days) and showed a 100% revival rate under favourable conditions. This is the first report that has quantified these VBNC cell subpopulations and compared their different resuscitation potentials.
The researchers were also able to identify these large coccoid VBNC cells in a seafood sample that had previously tested negative for V.parahaemolyticus, suggesting that conventional testing methods may not pick up dormant bacteria.
Enzymes hold the key to resuscitation
Dr Wagley was also able to compare the proteins in both subpopulations and found that most of the proteins which were significantly up- or downregulated were involved in metabolism.
Using advanced microscopy techniques, Dr Wagley has been able to identify and characterise dormant, VBNC bacterial populations.
Studies have shown that an enzyme called lactate dehydrogenase may be key. This enzyme breaks down lactate into pyruvate, a crucial step in several metabolic pathways. This suggests that lactate dehydrogenase is required by the cells to survive the dormant phase, as well as for reviving them once conditions become more favourable for survival.
The study showed that a lactate dehydrogenase protein was significantly upregulated in all subpopulations of VBNC cells. Deleting this protein caused the cells to enter the VBNC state much quicker than they normally would, suggesting that lactate dehydrogenase may play an important role in regulating the dormant VBNC state. It may do this in part due to its ability to combat oxidative stress, which occurs through factors such as environmental stressors.

Impact for seafood-borne diseases
Dr Wagley’s research has helped develop and standardise methods used to identify and quantify V.parahaemolyticus in seafood. This has the potential to reduce seafood-borne disease as it may be possible to detect subpopulations of the bacteria which were not previously considered. Understanding the relationship and transition between VBNC cells and cells able to grow and replicate is key to understanding about the incidence of potential gastrointestinal disease from the environment.
Going forwards, the group at Exeter will continue this work by further investigating the impact the VBNC state of V.parahaemolyticus may have on food safety as well as expanding the knowledge around this phenomenon.
Are VBNC bacterial cells in seafood still able to cause disease if ingested by humans?
This is not completely clear yet, but data in this study shows that when we collected them, the dormant VBNC cells didn’t cause disease in the larvae infection model we used. This suggests that VBNC cells are not virulent for this strain we tested. Once they start to wake up and revert to a culturable state they can cause disease again. There are different strains of V.parahaemolyticus that vary in their ability to cause disease and so more strains would need to be tested to check to see if this result is true for all V.parahaemolyticus strains.
References
- Wagley, S., Morcrette, H., Kovacs-Simon, A., Yang, Z. R., Power, A., Tennant, R. K., Love, J., Murray, N., Titball, R. W., & Butler, C. S. (2021). Bacterial dormancy: A subpopulation of viable but non-culturable cells demonstrates better fitness for revival. PLoS pathogens, 17(1), e1009194. Advance online publication. Available at: https://doi.org/10.1371/journal.ppat.1009194
10.26904/RF-134-1417
Research Objectives
Dr Wagley investigates how warming sea temperatures in the UK are affecting the increased incidence of Vibrio species.
Funding
- Biotechnology and Biological Sciences Research Council (BBSRC)
- Lyons Seafoods
Bio
Dr Sariqa Wagley carried out her PhD at the Centre for Fisheries and Aquaculture Science (Cefas) on Vibrio species in the environment. She has been working in the Biosciences department at the University of Exeter to understand host pathogen interactions at the molecular and cellular level in a number of bacteria that cause disease in humans.

Contact
Sariqa Wagley
Biosciences, College of Life and Environmental Sciences
Geoffrey Pope Building
University of Exeter
Exeter, Devon, EX4 4QD
UK

E: s.wagley@exeter.ac.uk
T: +44 1392 725177
Twitter: @sariqawagley
W: biosciences.exeter.ac.uk/staff/profile/index.php?web_id=sariqa_wagley









