An aortic aneurysm is a permanent dilation of the main blood vessel leaving the heart, creating a bulge which has the potential to rupture suddenly with deadly consequences. These aneurysms can occur at different locations, either above or below the diaphragm, and are thus termed either thoracic aortic aneurysms (TAA) or abdominal aortic aneurysms (AAA), respectively.
When a bulge becomes a bomb
Although it is unclear why aneurysms develop, there are several risk factors associated with this condition, including smoking and advanced age. They are also more prevalent in men than women. Since the disease has no medical therapy, a diagnosis can be devastating.
TAAs are the life-threatening
manifestation of Marfan’s disease![]()
Early research led by Dr Daugherty and Dr Cassis investigated the role of angiotensin II, a signalling molecule, in the development of arterial plaques. Dr Daugherty’s interest in aneurysms however, was sparked by the serendipitous discovery that AAAs were formed in a mouse model developed by his team to determine the effects of increased blood pressure on atherosclerosis. The model was developed to exhibit excessive blood lipid content (fat in the form of cholesterol). Early experiments showed that angiotensin II promoted plaque formation without increases in other measures, such as blood pressure. More interestingly, it significantly increased the occurrence of aortic aneurysms.
Blowing the field wide open
This discovery was further strengthened by the absence of any such effects in the control mice that do not express hyperlipidaemia (high levels of lipids in the blood). This led Dr Daugherty to conclude that high angiotensin II levels combined with hyperlipidaemia have “profound and rapid effects on vascular pathology”.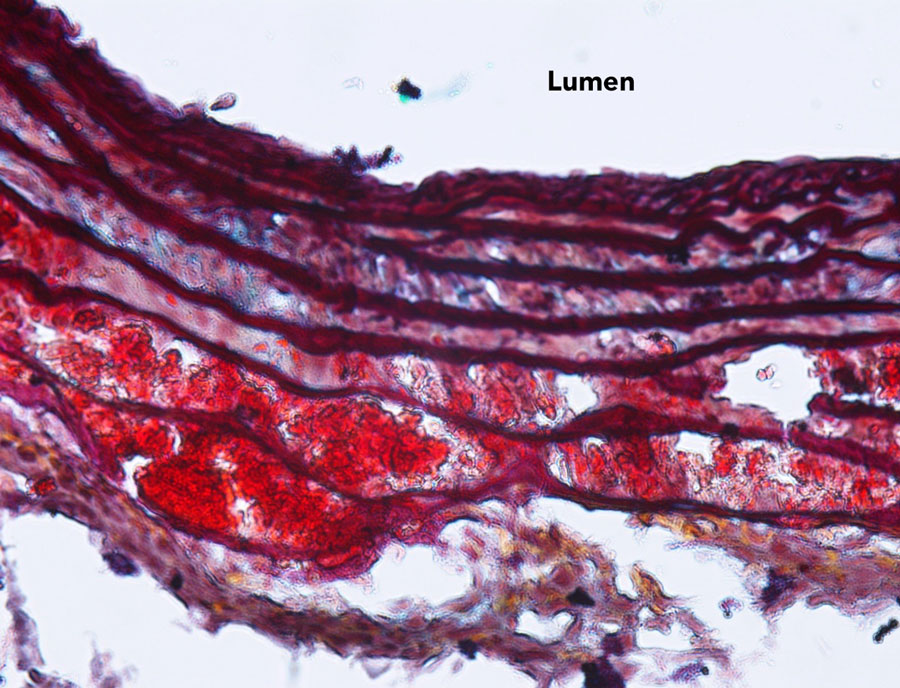
Identifying the detonator
Progress was made on this aspect of aneurysm development whilst examining the effects of chemokine receptors on atherosclerosis and AAA. In the same hyperlipidaemic mouse model, with CCR2 (a chemokine receptor) knocked out, instances of AAA and atherosclerosis were diminished. The research team observed that mice with active CCR2 also developed TAAs with markedly different pathological profiles when compared to AAA. Despite these differences, they also were diminished in CCR2-depleted mice thereby demonstrating a distinct role for chemokines in this vascular pathology.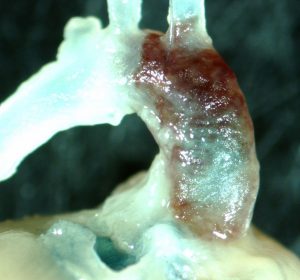
The highly contrasting pathologies between [these areas] are indicative of different mechanisms by which angiotensin II generates these diseases![]()
According to Dr Daugherty, another difference is in the ascending aorta (the thoracic part of the aorta which leads away from the top of the heart before curving back down to take freshly oxygenated blood to the major organs) the springy elastin proteins in the aortic wall are widely broken down, but there are interesting regional differences that may reflect the functional heterogeneity within the aorta. Medial dissection, a condition in which blood accumulates within the wall of the aorta, is also common. This same pathology occu
Disarming the bomb
Current therapies for both AAA and TAA rely on early diagnosis and surgical intervention, and failure to act on the problem can result in deadly rupture. As this condition is prevalent in Marfan syndrome, these patients are in particular need of therapies which can prevent the formation of aneurysms to reduce the risk of deadly consequences.
One class of drugs, currently used to treat hypertension (high blood pressure), shows promise as a treatment due to its blocking of the angiotensin II type 1 receptor. There is considerable debate among scientists in this field as to whether this is preferential to the alternative treatment which inhibits the formation of angiotensin II by blocking the angiotensin converting enzyme (ACE). This effect is seen because there is also an inhibitory effect of angiotensin II via the type 2 receptor, which appears to be beneficial in reducing many of the type 1 receptor’s effects. Dr Daugherty and Dr Sheppard are actively pursuing this concept.
The identification of similar signalling pathways in the aortic wall of patients with other syndromes where aortic aneurysm is a major risk factor, suggests that any treatment which is successful in one area may have broad action across these conditions. It is also true that there are potential risks with such treatments that disrupt the normal signalling pathways in a non-tissue-specific manner, but for Drs Daugherty and Sheppard defusing this ticking bomb for those with Marfan syndrome and other conditions is of prime importance.
I still remember the day that we found our first angiotensin II-induced aortic aneurysms! Rigorously-derived research data is a demanding discipline that requires the acquisition of high skill levels and the repetitive performance of experiments to ensure the results are statistically robust. One of the consequences of the need for rigour is that acquisition of new knowledge can be incremental. Hence it is exciting, and unusual, for an experiment aiming to provide one hypothesis, to provide great insight into another discipline. In this case, our excitement was augmented by the discovery of an animal model in a disease entity that has been greatly under-researched, despite its devastating effects on public health.
What impact have Mary Sheppard and the Marfan community had on your research?
A primary motivation for my research is the hope that it will directly alleviate this disease. The presence of Mary provides both myself, and my research team with a very palpable illustration of the impact of this disease, and heightens our sense of urgency to uncover new insights that may assist in development of new treatments.
Why are the different types of aneurysms so location specific?
This is a major unanswered question, whose answer would likely provide great insight into the disease process. One of the potential causes for the different locations is the complexity of the aorta. While the aorta has previously been considered a mere conduit for blood flow, it is now realised that there are major differences along the aorta that could impact aneurysm location. These include many differences such as flow patterns, extracellular matrix fibres, oxygen and nutrient delivery, and cell types that form the outside scaffold of the aorta. Presumably the location specificity is a consequence of one or a conglomeration of these factors.
A primary motivation for my research is the hope that it will directly alleviate Marfan syndrome![]()
What are the implications for this in research and treatment?
Elucidation of the basis for the regional specificity of aneurysms may provide insight into a mechanism that is effective and well-tolerated. Many of the currently proposed mechanisms have the potential to lack specificity. For example, drugs that inhibit the proteolysis of extracellular matrix have been proposed to benefit aneurysms, but this class of drugs is known to have side effects. However, if a unique mechanism could be determined in the aneurysmal region, it may provide the opportunity to inhibit a unique mechanism of action or develop a focal delivery mechanism to reduce effects outside of the aneurysm.
What are next steps in tackling TAA in Marfan syndrome?
There are several ongoing clinical trials testing whether it is beneficial to administer drugs that inhibit the ability of angiotensin II to stimulate one of its major receptors, AT1. The currently completed trials have provided a mixed message. However, they have all used a drug that is suboptimal to inhibit the action of angiotensin II. Some of the ongoing trials are using more effective drugs, and the outcome of these trials will be a major driver of the adoption of angiotensin receptor inhibition therapy, or illustrate the need to take another direction. At the moment, there is little consensus of an alternative mechanism.
Dr Daugherty’s research focuses on aortic aneurysmal diseases, particularly thoracic aortic aneurysms. Currently, little is known about the cause of these diseases and relevant therapeutics are largely inadequate. Dr Daugherty is looking to change this.
Funding
National Institutes of Health (NIH)
Collaborators
- Debra Rateri
- Lisa Cassis
- Mary Sheppard
Bio
 Dr Alan Daugherty completed a BSc in Pharmacology at Sunderland Polytechnic and received his PhD and DSc from the University of Bath. He moved to Washington University in St. Louis for fellowship training and was subsequently appointed to the faculty at Washington University. In 1997, he moved to the University of Kentucky.
Dr Alan Daugherty completed a BSc in Pharmacology at Sunderland Polytechnic and received his PhD and DSc from the University of Bath. He moved to Washington University in St. Louis for fellowship training and was subsequently appointed to the faculty at Washington University. In 1997, he moved to the University of Kentucky.
Contact
Dr Alan Daugherty, PhD
B-243 BBSRB
Saha Cardiovascular Research Center
University of Kentucky
Lexington, KY
40506 – 0509
USA
E: alan.daugherty@uky.edu
T: +1 859 323 3512
W: https://cvrc.med.uky.edu/users/adaugh









