Mechanical ventilation is a crucial intervention used when a person is unable to breathe on their own. However, mechanical ventilation is often not in synchrony with the patient’s own breathing. Drs Jennifer Beck and Christer Sinderby, PhD physiologists at St Michael’s Hospital, University of Toronto, have developed a novel technology known as neurally adjusted ventilatory assist, or NAVA. Clinicians Dr Howard Stein, Kimberly Firestone, and Dr Sherry Courtney, among others, have published several findings utilising this technology. NAVA directly senses a patient’s respiratory effort, then provides the correct amount of ventilation support, breath-by-breath. This increases the patient’s comfort, decreases the need for sedation, and may decrease associated pathologies.
Mechanical ventilation is a critical intervention used when a patient is unable to breathe on their own. Ventilation can be required for several reasons. A person may be admitted to hospital because they have pneumonia and are having trouble breathing. Ventilation assists breathing and allows the body to focus all its strength on an immune response. Ventilation can also be used for individuals who have suffered trauma to their nervous system, preventing them from being able to breathe themselves. Individuals who are unconscious, for example if they have taken a drug overdose, could also require help breathing. A more contemporary reason for needing mechanical ventilation occurs when individuals become ill with COVID-19, which develops into life-threatening pneumonia – this prevents an adequate supply of oxygen to the body. Regardless of the reason for mechanical ventilation, the objective is the same: to deliver oxygen into the lungs and reduce the amount of carbon dioxide in the body. This is achieved by a ventilator delivering oxygen into the patient’s lungs via an endotracheal tube that is inserted into the windpipe via the mouth or nose. Another vital role for a ventilator is to provide positive end-expiratory pressure – or PEEP. PEEP prevents the patient’s air sacs in the lungs from collapsing due to damaged lung tissue.

In principle, mechanical ventilation should accurately mimic a person’s natural breath so that oxygen is delivered in the best way. In practice, however, this does not always occur. Tidal volume is the normal amount of air which a person inhales and exhales without extra effort. In the simplest form of mechanical ventilation, the tidal volume is set by the medical staff. If this tidal volume is not in sync with the patient’s own respiratory cycle, severe problems can follow due to a poor patient-ventilator interaction. The respiratory mismatch causes the patient to ‘fight’ ventilation. To prevent this, sedatives or muscle paralytics are administered. This can cause over-ventilation, deterioration of the muscles or a bad reaction to medication.
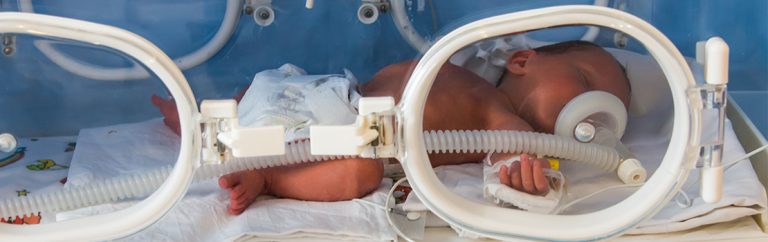
A further consideration is the ventilation of young infants. Mechanical ventilation has dramatically increased the survival rate of infants who are born with respiratory failure, but the side-effects can be very dangerous. In the short term, ventilating infants can increase the risk of damaging the airways, air leaks in the lung tissue or infection; in the long term, it increases the risk of bronchopulmonary dysplasia where the lungs fail to develop properly and long-term oxygen is required.
In NAVA, the patient tells the ventilator how it should help rather than the ventilator controlling the breath of the patient.
So what is the solution? In an ideal world, we would have machinery which could measure our breathing and modulate the ventilation accordingly. Our breathing would be measured using the respiratory centre’s output located in the brainstem, or the phrenic nerve activity which controls the contraction of the muscle we use for breathing. However, neither of these options are viable. It is currently not feasible to directly measure the output of either in humans. But what about focusing on the muscle we use for breathing, known as the diaphragm?

Source: depositphotos.com
The diaphragm is the major respiratory muscle and is located just below the lungs. When an individual breathes in, the diaphragm contracts and flattens, causing the chest cavity to expand. This creates a vacuum that pulls air into the lungs. When an individual breathes out, the diaphragm relaxes and air is forced out of the deflated chest cavity. The contraction of the diaphragm is directly related to the activity of the phrenic nerve. Up until now, measuring the contraction of the diaphragm was also not an option, as electrical activity of organs such as the heart polluted any signal recorded. This is no longer the case as improvements in technology mean we are able to record a pure, unpolluted diaphragmatic electrical signal.
Neurally Adjusted Ventilatory Assist
Neurally adjusted ventilatory assist – or NAVA – was invented by Dr Jennifer Beck and Dr Christer Sinderby. It is a revolutionary way of delivering mechanical ventilation. In NAVA, an array of miniaturised sensors is attached to the nasogastric tube typically used to feed patients. This allows the sensors to sit in the patient’s esophagus and stomach. The sensors pick up the breathing signals from the diaphragm which are amplified and delivered to a computer that filters out any background electrical activity. This signal represents the diaphragmatic activity, called Edi, and is used to modulate the amount of ventilation delivered to the patient. A “NAVA level” is set by the clinician and assists each diaphragmatic signal (see figure 1). In essence, in NAVA the patient tells the ventilator how it should help rather than the ventilator controlling the breath of the patient. This is important not only for the comfort of the patient but for reducing ventilation-associated pathology. NAVA was approved by the Food and Drug Administration in 2007 and there is much evidence of its efficacy in patients of all ages, from tiny premature infants to adults.

The efficacy of NAVA in ventilating infants
As mentioned previously, ventilation of infants can be damaging to their short-term and long-term health. Several researchers have investigated the potential of NAVA in improving mechanical ventilation in infants and have collected evidence demonstrating that NAVA should have a role in a pediatric setting. In 2009 Beck, Sinderby and collaborators were able to show that using the diaphragmatic signal to control ventilation with NAVA improved patient-ventilator interaction when ventilating premature babies with differing lung diseases. This allows patients to determine when the ventilator breath starts, the size of the breath delivered and when to terminate the breath.

Source: wikipedia.org/wiki/Respiratory_system
In 2015, Firestone, Stein and associates demonstrated that NAVA can accurately assist the breathing at varying NAVA levels. Higher NAVA levels allow the work of breathing to shift from the diaphragm to the ventilator. Infants decrease their breathing effort if the NAVA level is too high. The point at which this occurs is known as the breaking point and is different for everyone. In 2020 Firestone, Stein and associates also showed that premature neonates are able to regulate each breath size and these typically fall within the range usually provided by the clinician using conventional ventilation. An extremely important advantage of NAVA is that it can be used during non-invasive ventilation (NIV), that is, ventilation with a mask or nasal cannula and no endotracheal tube in the airway. No other form of non-invasive ventilation can provide totally synchronised breaths in this fashion. NIV has been used to decrease the damaging effects of ventilation in infants by being less physically intrusive. In 2018 Courtney and co-workers found that NIV NAVA significantly reduced the work of breathing in piglets with both healthy and diseased lungs.
The group was able to show that NAVA and recording of the diaphragmatic signal was suitable when ventilating premature babies with differing
lung diseases.
Taken together, the evidence for the use of NAVA both in infants and adults is overwhelmingly positive. NAVA allows ventilation to adapt to individual patients, different age groups and when ventilation is required for different reasons. NAVA allows each patient to breathe at their own rate and depth, comfortably and with decreased or even no sedation. NAVA ventilation represents an exciting leap forward in the technology available for helping patients do the most important thing that humans do: breathe.

What inspired you to conduct this research?
Dr Courtney: From my earliest days as a researcher, I have been interested in exploring methods of assisted ventilation. NAVA was a clear advance in support of breathing and thus I wished to learn more about it.
Dr Stein and Ms Firestone: Our research aims were to explore the possibility of reducing time on the ventilator and lung injury for premature neonates.
Dr Beck: The interaction between a breathing baby and the respirator is not synchronous; this can be uncomfortable and dangerous. My goal was to develop a method that would allow clinicians to detect this asynchrony, and to fix the problem.
Dr Sinderby: My goal was to integrate the patient’s neural control, and neural monitoring into the treatment of mechanical ventilation. I was looking for a way to treat the lungs but also allowing the patient to autonomously control and protect him/herself.
References
- Sinderby, C., Navalesi, P., Beck, J., Skrobik, Y., Comtois, N., Friberg, S., Gottfried, S.B., and Lindström, L. (1999). Neural control of mechanical ventilation in respiratory failure. Nature Medicine, 5(12), 1433-6.
- Beck, J., Reilly, M., Grasselli, G., Mirabella, L., Slutsky, A.S., Dunn, M.S., and Sinderby, C. (2009). Patient-ventilator interaction during neurally adjusted ventilatory assist in low birth weight infants. Pediatric Research, 65(6), 663-8.
- Firestone, K., Fisher, S., Reddy, S., White, D.B., and Stein, H.M. (2015). Effect of changing NAVA levels on peak inspiratory pressures and electrical activity of the diaphragm in premature neonates. Journal of Perinatology, 35, 612-6.
- Jones, M.L., Bai, S., Thurman, T.L., Holt, S.J., Heulitt, M.J., and Courtney, S.E. (2018). Comparison of work of breathing between noninvasive ventilation and neurally adjusted ventilatory assist in a healthy and a lung-injured piglet model. Respiratory Care, 63(12), 1478-84.
- Protain, A., Firestone, K.S., McNinch, N., and Stein, H. (2020). Evaluating Peak Inspiratory Pressures and Tidal Volume in Premature Neonates on NAVA ventilation. European Journal of Pediatrics. Available at: https://doi.org/10.1007/s00431-020-03728-y
10.26904/RF-134-6669
Research Objectives
Dr Beck, Dr Sinderby and collaborators have devised a new method of assisting patients on mechanical ventilation: Neurally Adjusted Ventilatory Assist (NAVA).
Funding
- Getinge
- The RS McLaughlin Fund
- The Arkansas Children’s Research Institute
Collaborators
- David Matlock
- Norm Comtois
- Michelle Jones
Bio

Dr Jennifer Beck developed a method to accurately measure the electrical activity of the diaphragm, now used in the critical care setting to monitor patients and control mechanical ventilation. Dr Beck is co-inventor of NAVA. Her current work is focused on new ventilation methods for preterm newborns (“Helping Babies Breathe Better”).

Dr Christer Sinderby’s work focuses on control of breathing during mechanical ventilation in acute respiratory failure. He also does research on new techniques and methods to support the breathing of critically ill patients. Dr Sinderby is co-inventor of NAVA.

Dr Howard Stein is Director of the Neonatal Intensive Care Unit (NICU) at ProMedica Ebeid Children’s Hospital in Toledo, Ohio. He is a Professor of Pediatrics at the University of Toledo College of Medicine and Life Sciences. As an expert on NAVA, he is doing clinical trials utilising NAVA in the NICU.

Dr Sherry Courtney is Professor of Pediatrics and Director of Clinical Research in the Section of Neonatology, University of Arkansas for Medical Sciences, Little Rock, Arkansas. She trained at the University of Kansas School of Medicine and Children’s Mercy Hospital in Kansas City, Missouri.

Ms Kimberly Firestone MSc, RRT is the Neonatal Respiratory Outreach Clinical Liaison for the Neonatal Intensive Care Unit at Akron Children’s Hospital. As a respiratory therapist she was responsible for coordinating the efforts to bring NAVA technology into their unit.
Contact
Arkansas Children’s Hospital
One Children’s Way, Slot 512
Little Rock, AR 72202, USA
E: scourtney@uams.edu
T: +1 501 364 1028