The therapeutic and research benefits of electrically stimulating tissues are long established, from pacemakers and treatments for movement disorders such as Parkinson’s disease to the identification of causal relationships between brain regions and functional responses. Initially, these were limited to direct electrical stimulation, by inserting fine electrodes into heart or brain tissue and delivering an appropriate current.
This approach is clearly not without its problems. Electrode implantation itself poses risks to the individual, and with the advent of magnetic resonance imaging (MRI), further complications were created. During MRI scanning, the ‘antenna effect’ induces an electrical current in the tip of the electrode which dissipates as heat; potentially causing irreversible damage to the tissue surrounding it.
Done with magnets
Transcranial magnetic stimulation (TMS) offered the potential to overcome these issues by firstly not requiring invasive surgical procedures. Stimulation is effected by placing a magnetic coil in close proximity to the target area of the brain; varying the magnetic field causes an electric current to flow in a small area of the brain through electromagnetic induction. It has been useful diagnostically in a range of diseases which relate to the motor cortex, such as stroke, multiple sclerosis, and motor neuron diseases, it has also been used to treat neuropathic pain (pain caused by damage or disease affecting the region of the brain which deals with bodily sensations).
One other advantage of TMS is that it can be used in conjunction with functional MRI to improve our understanding on how TMS stimulates the brain, and its probes can be built into special MRI-safe headsets and stimulation delivered during scanning. The major disadvantage is its spatial resolution, fine targeting of the stimulation is difficult due to the properties of magnetic fields and the engineering limits of electromagnetic coil design.
An imprecise science
‘Bleed over’ into nearby areas of the brain mean that its use as a research tool is limited. However, it has been successfully deployed to interrogate motor pathways, language organisation and for pre-surgical evaluation of patients. Its use as a mapping tool to assess how neural pathways are disrupted in brain tumour development, cerebral palsy and epilepsy are perhaps the most clinically relevant uses of this technology, providing vital information to physicians on the progress of these diseases.
The therapeutic uses of both TMS and direct electrical stimulation are now beginning to overlap, the networked nature of the brain means that there are nodes in the network which have the capacity to impact multiple disease expressions. These can be stimulated either directly, as is more usual for deep tissue stimulation, or by TMS which is able to operate only at relatively shallow regions due to the method of delivery.
The therapeutic uses of both TMS and direct electrical stimulation are now beginning to overlap.
Seeing the light
All the cells which responded to light stimulation also responded to μMS. Dr Bonmassar described the results in the journal Nature as, “The amplitude and kinetics of individual biphasic waveforms [where the response cycles above and below the baseline value] were nearly identical to that of action potentials elicited in response to light stimuli, strongly suggesting that the biphasic waveforms were in fact action potentials [the wave of electrical energy that underlies a nerve impulse].” These exciting results laid the foundations for further refinement of the process in vitro and demonstrated the real potential of the product he had designed.
The team continued to investigate the mechanism and salient features of the stimulation effects; showing that the orientation of the coil and the magnitude of the initiating current, amongst other parameters, were fundamental to the response obtained. This result demonstrates a level of specificity and tuning which could prove vital for both the clinical and research applications of the technology.
Dr Bonmassar designed the experiments carefully. “Potential contributions from several non-magnetic factors including leaking electrical current, heating of tissue, and mechanical vibration were all eliminated,” he said, “allowing us to conclude that small magnetic fields can elicit action potentials.” He goes on to describe the effect this will have on the spatial resolution of magnetic stimulation as akin to the leap from low-field MRI technology to the ultra-high field MRI (currently not extensively deployed for clinical use).
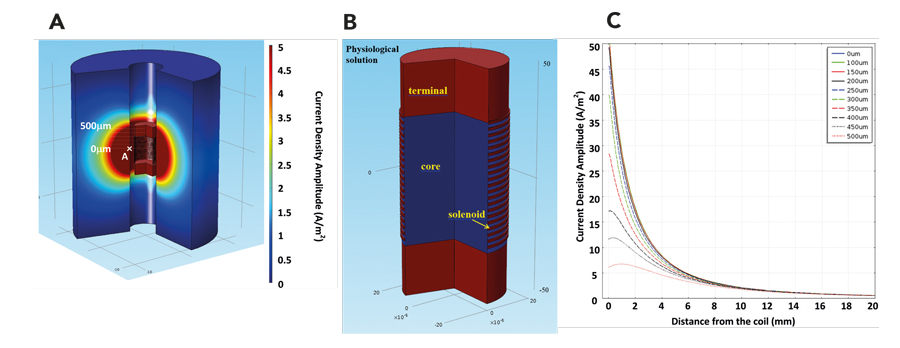
The key to achieving this is adapting the μMS technology to create the first generation of miniaturised TMS probes (μTMS) allowing for ultra-focal non-invasive brain stimulation. Advances in micro-fabrication allow for long electromagnetic coils to be created in rings as small as a few millimetres across. The length of the coil is vital to producing a field with sufficient strength to penetrate the skull while maintaining a low resistance ensures that heat generation remains low enough to prevent scalp discomfort.
The next step for Dr Bonmassar and his team of collaborators and researchers is to move to first-in-human trials of the technology. This research will demonstrate μTMS safety and reliability for clinical use as well as give them the opportunity to compare its mapping abilities to currently approved TMS systems. Dr Bonmassar believes that these early tests will clearly demonstrate the superior spatial precision and resolution of μTMS over traditional systems.
Micro-magnetic stimulation is an emerging technology with great promise to revolutionise therapeutic stimulation of the human nervous system.
Focus on the future
The team already have extensive modelling results to direct coil design, indicating that the μTMS coils produce a substantially more focused stimulation than traditional TMS probes while using a much smaller current. Dr Bonmassar believes this is due to micro coils being capable of eliciting action potentials at lower field thresholds due to the higher spatial gradient (their small size means a sharper decline in signal with distance).
“Micro-magnetic stimulation is an emerging technology with great promise to revolutionise therapeutic stimulation of the human nervous system,” says Dr Bonmassar, “Our project is adapting this novel technology to develop the first generation of μTMS probes.” He is confident that as they push this technology forward, “it will enable for the first time, ultra-high resolution non- invasive stimulation of the human brain with applications across therapeutic and exploratory neuroscience.”
In clinical practice, cortical mapping of language, as well as the systematic exploration of the motor cortical outputs can be beneficial for the pre-surgical evaluation of patients in order to characterise eloquent cortex whilst reducing the need for intraoperative evaluation. In addition to the highly improved focality, which allows for precise selection of a stimulation site, the focal simulation also reduces extraneous activation of non-targeted brain areas. Furthermore, the significantly reduced size of µTMS elements allows integrating them into multi-channel conformal head arrays for simultaneous multi-focal stimulation (e.g., a TMS helmet will be finally feasible). In the realm of network stimulation, the ability to apply TMS simultaneously in multiple sites to inhibit certain nodes while facilitating others will introduce a significant leap in the study of altered brain networks in psychiatric and neurological disorders.
References
- Bonmassar G. (2012). ‘Microscopic magnetic stimulation of neural tissue’. Nature Communications, Vol.3, No. 921, DOI: 10.1038/ncomms1914.
- Bonmassar G. (2014). ‘Optimizing Microscopic Magnetic Fields for Neuronal Stimulation’. International Journal of Bioelectromagnetism, Vol. 16, No. 1, pp. 1-31. http://www.ijbem.org/volume16/number1/ijbem_vol16_no1_pp1-31.pdf.
- Najib U, Bashir S, Edwards D, Rotenberg A, and Pascual-Leone A. (2011). ‘Transcranial brain stimulation: clinical applications and future directions’. Neurosurgery clinics of North America, Vol. 22, pp. 233-251.
- Wu A. D, Fregni F, Simon D. K, Deblieck C, and Pascual-Leone A. (2008). ‘Noninvasive brain stimulation for Parkinson’s disease and dystonia’. Neurotherapeutics, Vol. 5, pp. 345-361.
- Shirota Y, Ohtsu H, Hamada M, Enomoto H, Ugawa Y, and Research Committee on rTMS Treatment of Parkinson’s Disease. (2013). ‘Supplementary motor area stimulation for Parkinson disease: a randomized controlled study’. Neurology, Vol. 80, pp. 1400-1405.
Project Narrative Micro-magnetic stimulation (μMS) is an emerging technology with a great promise to revolutionise therapeutic stimulation of human nervous system. In this project, they will adapt this novel technology to develop the first generation of miniaturised transcranial magnetic stimulation (μTMS) probes. The outcome will enable for the first time, ultra-high resolution non- invasive stimulation of the human brain with applications in therapeutic and exploratory neuroscience.
Funding
NIH-MIMH R01MH111875
Collaborators
- Golestanirad L, Zhao L, Press D, Yang S, Pascual-Leone , Athinoula A Martinos Center, Department of Radiology, Massachusetts General Hospital, Boston, MA.
- Department of Mechanical Engineering, Massachusetts Institute of Technology, Cambridge, MA.
- Department of Neurology, Beth Israel Deaconess Medical Center, Boston, MA.
Bio
 Giorgio Bonmassar, PhD is an Associate Professor of Radiology at Massachusetts General Hospital and Harvard Medical School with expertise of over twenty years in electrophysiology systems measurements simultaneous to MRI, including a set of microelectrodes that are truly MRI-invisible that led to a Science article. The feasibility of Micro-magnetic stimulation (μMS) implanted in deep brain structures was first shown by his group of being capable of delivering therapeutic stimulation with effects analogous to the state-of-the-art deep brain stimulation.
Giorgio Bonmassar, PhD is an Associate Professor of Radiology at Massachusetts General Hospital and Harvard Medical School with expertise of over twenty years in electrophysiology systems measurements simultaneous to MRI, including a set of microelectrodes that are truly MRI-invisible that led to a Science article. The feasibility of Micro-magnetic stimulation (μMS) implanted in deep brain structures was first shown by his group of being capable of delivering therapeutic stimulation with effects analogous to the state-of-the-art deep brain stimulation.

Contact
Prof Giorgio Bonmassar, PhD
Associate Professor
AA. Martinos Center
Massachusetts General Hospital
Harvard Medical School
Building 75, Third Ave
Charlestown, MA 02129
USA
E: Giorgio.Bonmassar@mgh.harvard.edu
T: +1 617 726 0962
W: https://www.nmr.mgh.harvard.edu/user/5622
W: https://connects.catalyst.harvard.edu/Profiles/display/Person/26317









