Viruses can cause some of the most fatal diseases on Earth. The infectious nature of viruses enables them to spread rapidly throughout populations and cause global epidemics. For example, one of the most prevalent viral diseases are caused by dengue viruses, which originated in Africa and South Asia. This mosquito-transmitted viral infection has increased in incidence over the last 50 years and spread to over 100 countries. Alarmingly, around 390 million infections occur per year, of which over 90 million manifest clinical symptoms, and approximately half of the global population are at risk.
Viruses hijack healthy host cells, exploiting resources and reproductive machinery for rapid replication. However, a major consequence of speedy reproduction is the increased risk of random genetic mutation, which can result in a myriad of unique viral strains. Therefore, viruses are very difficult to treat. Vaccines that target a specific viral strain could become ineffective in the long-term. This is because viruses could genetically alter to become resistant to the antibodies raised by a vaccine. Ultimately, a conflict exists between humans and viruses, with each side battling to overcome obstacles created by the opposition.

How do viruses enter cells?
Essentially, viruses consist of two key elements – a nucleic acid molecule and a protein coat. Some of the these proteins, glycoproteins, are used to gain entry into healthy cells by binding to its specific receptor, found on the cell membrane. Additionally, interactions with coreceptors and various attachment factors enhance infection. When the viral glycoprotein binds to its receptor, a process is triggered in which the host and viral membranes fuse and the genetic material of the virus is delivered into the cell. Professor Choe has investigated these viral glycoproteins and cellular receptors for a range of different viruses.
Chemokine receptors are needed for HIV infection
Professor Choe started her journey of virus research by studying Human Immunodeficiency Virus (HIV). HIV is one of the most severe global health issues having claimed approximately 35 million lives. This devastating virus infects healthy cells including T lymphocytes, dendritic cells and macrophages (cells of the immune system), and the cells in the central nervous system. These diverse range of cells have one factor in common – they all express the protein, CD4, on the cellular membrane. CD4 acts as a receptor for the HIV, and upon binding a virus, it helps internalising the virus into the cell. Professor Choe identified two key chemokine receptors, CCR3 and CCR5, which are also required to facilitate HIV infection. These chemokine receptors are expressed on immune cells and help the cells to migrate to infection sites. HIV then infects these immune cells, gathered at the infection sites, utilising chemokine receptors expressed on them. The team showed this by manipulating HIV-resistant cells to become susceptible to HIV infection by expressing CCR5 or CCR3 coreceptor as well as CD4 receptor.
Professor Choe identified two key chemokine receptors, CCR3 and CCR5, facilitate HIV infection as coreceptors.
Angiotensin converting enzyme 2 supports SARS virus entry into cells
When Severe Acute Respiratory Syndrome (SARS) virus broke out and wreaked the havoc in 2003, Professor Choe together with long-term collaborator Professor Michael Fazan joined the SARS crusade, and identified the SARS virus receptor, angiotensin converting enzyme 2 (ACE2), at a lightning speed. SARS was first detected in February 2003, and the Farzan and Choe laboratories identified ACE2 by the summer of the same year. The identification of ACE2 is very important not only for its own sake but also for the scientific confirmation of palm civets as the immediate source of SARS virus. The same Choe/Farzan team discovered that the deadly stain of SARS virus derived from 2003 outbreak could utilise both civet and human ACE2 molecules to efficiently infect cells, but the mild strain isolated from the subdued outbreak in the following year efficiently used civet ACE but not human ACE. This discovery is crucial, because it explains at a molecular level why and how 2003 strains of SARS virus could easily jump to humans while 2004 strains could not, and helps to predict which animals could or could not be the carrier for SARS viruses. Later, together with other precautionary measures, eliminating all palm civet trading in animal markets contributed to bringing an end to SARS outbreaks.
This ground-breaking research inspired Professor Choe to investigate the processes by which other viruses enter and manipulate cells, for example New World arenaviruses including Machupo, Guanarito, Junin and Sabia viruses. These viruses cause actue haemorrhagic fever in humans with mortality rates as high as 30%. Research conducted by the team showed that transferrin receptor 1 (TfR1) had a high affinity for the entry glycoprotein of Machupo virus. The team manipulated hamster cells, which are not susceptible to Machupo virus, to express human TfR1, and found that these cells became infected by Machupo virus. This phenomenon was observed by human TfR1, but not by a closely-related protein, TfR2. The team also found that human TfR1 was used by all other deadly New World arenaviruses to invade cells. Furthermore, the team investigated various strategies that could be used to inhibit arenavirus infection. Iron depletion is known to enhance synthesis of the TfR1 protein. Therefore, Professor Choe showed that iron supplementation decreased the efficiency of infection of hemorrhagic-fever New World arenaviruses such as Junin and Machupo. Additionally, the team showed that use of TfR1 antibodies can effectively block the virus-receptor interaction, meaning that the viral glycoprotein cannot bind to this receptor and therefore cannot enter the cells.
Apoptotic mimicry
More recently, the Choe laboratory was involved in several significant studies that focus on the so-called ‘phosphatidylserine (PS) receptors’. PS plays a key role in the process of apoptosis (programmed cell death). In healthy cells the enzymes, called flippases, activey ensure that PS is confined to the inner-side of the cellular membrane. However, during apoptosis, flippases are inactivated and the enzymes scramblases act upon PS, causing this phospholipid to be exposed on the outer-side of the cellular membrane. This exposed PS on apoptotic cells acts as an ‘eat-me’ signal; it binds to PS receptors expressed on macrophages (a type of immune cells) and induces them to engulf apoptotic cells.
The Choe laboratory have shown that many enveloped viruses utilise these PS receptors to enter cells. This phenomenon is described as ‘apoptotic mimicry’, because viruses expose PS on their membrane surface, mimicking apoptotic cells. The membrane of enveloped viruses is derived from the cellular membrane, and because viruses do not contain flippases, PS located on the inner side of the virus membrane flops to outer side with time. These viruses, ‘disguised’ as apoptotic bodies, are engulfed by macrophages and other cells that express PS receptors, resulting in the infection of those cells.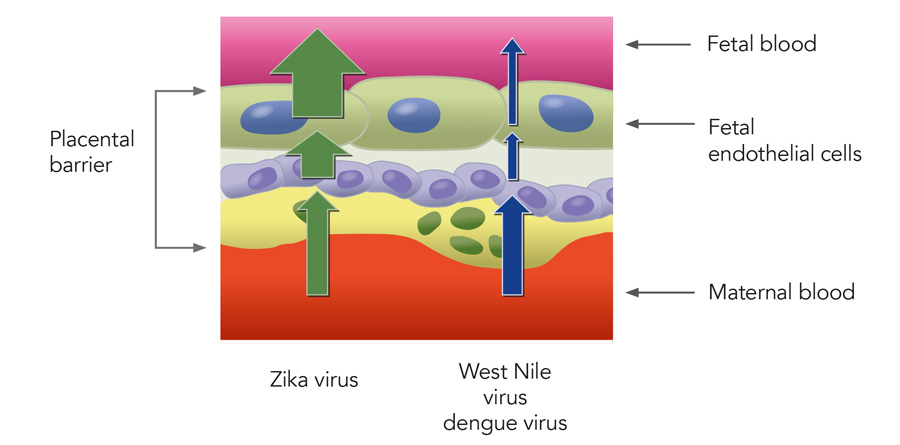
The Choe laboratory have shown that many enveloped viruses utilise these PS receptors to enter cells.
AXL and Zika virus interaction
Currently, Professor Choe and her colleagues are investigating another PS receptor, AXL, which specifically aids Zika virus infection, a flavivirus, although all flaviviruses are very similar in their genetic material and shape. Interestingly, the team found that AXL does not support the entry of other flaviviruses. AXL is found on fetal endothelial cells, therefore the Zika virus can interact with AXL and infect fetal endothelial cells, whereas other flaviviruses such as West Nile and dengue viruses cannot. This observation is of great importance because it explains how Zika virus can breach the placental barrier and spread via fetal circulation to the brain. The infection of the fetal brain with Zika virus can be life-threatening and cause the devastating condition, microcephaly, characterised by a smaller than normal head size, potentially resulting in physical and/or mental retardation of the babies born with it.
Future research
Overall, the research of Professor Choe and her team has greatly helped our understanding of how different viruses infect healthy cells by using a range of host and viral proteins and exploiting a wide range of biochemical, biophysical and virological techniques. However, there are many questions that remain unanswered that the Choe laboratory aims to explore: I) Do flaviviruses, including Zika virus, actively enhance PS incorporation into the virion membrane, thereby ensuring efficient infection of neighbouring cells? ii) Why Zika virus, but not other flaviviruse, uses AXL and cross the placental barrier?, and iii) How does the use of the PS receptors help the transmission from the vector to the host? Investigating these essential questions will take the team one step further to developing effective treatments to fight against these devastating viruses.
After we identified CCR5 as the major coreceptor for HIV-1 in 1996, researchers and companies worked together to find small-molecule CCR5 inhibitors, and they eventually found effective inhibitors that are currently in human use. This process takes a long time, not only because it is difficult to find effective inhibitors but also because it takes many years to evaluate them in clinical trials; the first CCR5 inhibitor was approved by FDA in 2007. Nonetheless, when we know which molecule is used by a virus, we can attempt to block the interaction between the virus and molecule.
References
https://www.scripps.edu/research/faculty/choe
https://www.scripps.edu/news/press/2012/20121213flappts.
https://projectreporter.nih.gov/project_info_description
http://www.who.int/mediacentre/factsheets/fs103/en/
http://www.who.int/denguecontrol/disease/en/
http://www.who.int/mediacentre/factsheets/fs360/en/
- Choe, H., Farzan, M., Sun, Y., Sullivan, N., Rollins, B., Ponath, P.D., Wu, L., Mackay, C.R., LaRosa, G., Newman, W. and Gerard, N., Sodroski J. 1996. The β-chemokine receptors CCR3 and CCR5 facilitate infection by primary HIV-1 isolates. Cell, 85(7), pp.1135-1148.
- Li, W., Moore, M.J., Vasilieva, N., Sui, J., Wong, S.K., Berne, M.A., Somasundaran, M., Sullivan, J.L., Luzuriaga, K., Greenough, T.C. and Choe, H., Farzan M. 2003. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature, 426(6965), p.450.
- Radoshitzky, S.R., Abraham, J., Spiropoulou, C.F., Kuhn, J.H., Nguyen, D., Li, W., Nagel, J., Schmidt, P.J., Nunberg, J.H., Andrews, N.C. and Farzan, M., Choe. H. 2007. Transferrin receptor 1 is a cellular receptor for New World haemorrhagic fever arenaviruses. Nature, 446(7131), p.92.
- Jemielity, S., Wang, J.J., Chan, Y.K., Ahmed, A.A., Li, W., Monahan, S., Bu, X., Farzan, M., Freeman, G.J., Umetsu, D.T. and DeKruyff, R.H., Choe H. 2013. TIM-family proteins promote infection of multiple enveloped viruses through virion-associated phosphatidylserine. PLoS pathogens, 9(3), p.e1003232.
- Richard, A.S., Zhang, A., Park, S.J., Farzan, M., Zong, M. and Choe, H. 2015. Virion-associated phosphatidylethanolamine promotes TIM1-mediated infection by Ebola, dengue, and West Nile viruses. Proceedings of the National Academy of Sciences, 112(47), pp.14682-14687.
Professor Choe and her team study how the TIM family of phosphatidylserine receptors promote infections of a wide range of enveloped viruses including filoviruses and flaviviruses such as West Nile, dengue and Zika viruses. They are continuing with their efforts to identify and characterise host factors, which modulate virus infection, and use their insight to develop strategies to inhibit viral replication.
Funding
NIH
Bio
 Hyeryun Choe, Professor in the Department of Immunology and Microbiology at Scripps-FL received her PhD degree from Pennsylvania State University. Since then, she identified or involved in identifying receptors for three important viruses: HIV-1, SARS virus and New World hemorrhagic fever viruses. Before joining Scripps-FL, she was Associate Professor at Harvard Medical School, Boston, MA, USA.
Hyeryun Choe, Professor in the Department of Immunology and Microbiology at Scripps-FL received her PhD degree from Pennsylvania State University. Since then, she identified or involved in identifying receptors for three important viruses: HIV-1, SARS virus and New World hemorrhagic fever viruses. Before joining Scripps-FL, she was Associate Professor at Harvard Medical School, Boston, MA, USA. Audrey Stephanie Richard studied molecular and cellular biology at University of Lille 2, Lille, France, where she received her PhD degree. She is currently working with Prof Choe as Staff Scientist at Scripps-FL, and played an essential role in discovering the crucial difference between Zika virus and other closely-related flaviviruses.
Audrey Stephanie Richard studied molecular and cellular biology at University of Lille 2, Lille, France, where she received her PhD degree. She is currently working with Prof Choe as Staff Scientist at Scripps-FL, and played an essential role in discovering the crucial difference between Zika virus and other closely-related flaviviruses. Contact
Hyeryun Choe, PhD
Department of Immunology and Microbiology
The Scripps Research Institute
130 Scripps Way
Jupiter, Florida 33458, USA
E: hchoe@scripps.edu
T: +1 561 228 2440
W: www.scripps.edu/research/faculty/choe









